The concept of electron geometry is essential in understanding molecular shapes, focusing on the arrangement of electron groups around a central atom. Electron groups include both lone pairs and bonding pairs, which are treated similarly in this context. The number of electron groups can be 2, 3, or 4, each leading to distinct geometrical arrangements.
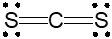
When there are 2 electron groups, the geometry is classified as linear. This can be visualized as two points connected in a straight line, emphasizing the simplicity of the arrangement.
For 3 electron groups, the geometry is termed trigonal planar. The prefix "tri-" indicates three, which helps in recalling this geometry. In this case, the arrangement can consist of either three surrounding atoms or two surrounding atoms with one lone pair.
When there are 4 electron groups, the geometry is described as tetrahedral. The prefix "tetra-" signifies four, making it easier to remember that this geometry involves four electron groups. This arrangement can include four surrounding atoms, two surrounding atoms with two lone pairs, or three surrounding atoms with one lone pair.
In summary, the electron geometries corresponding to 2, 3, and 4 electron groups are linear, trigonal planar, and tetrahedral, respectively. Understanding these geometries is crucial for predicting molecular shapes and behaviors in chemical reactions.