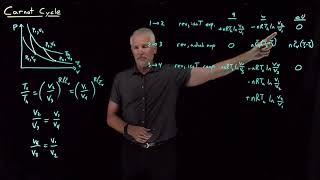Back
Back23. The Second Law of Thermodynamics
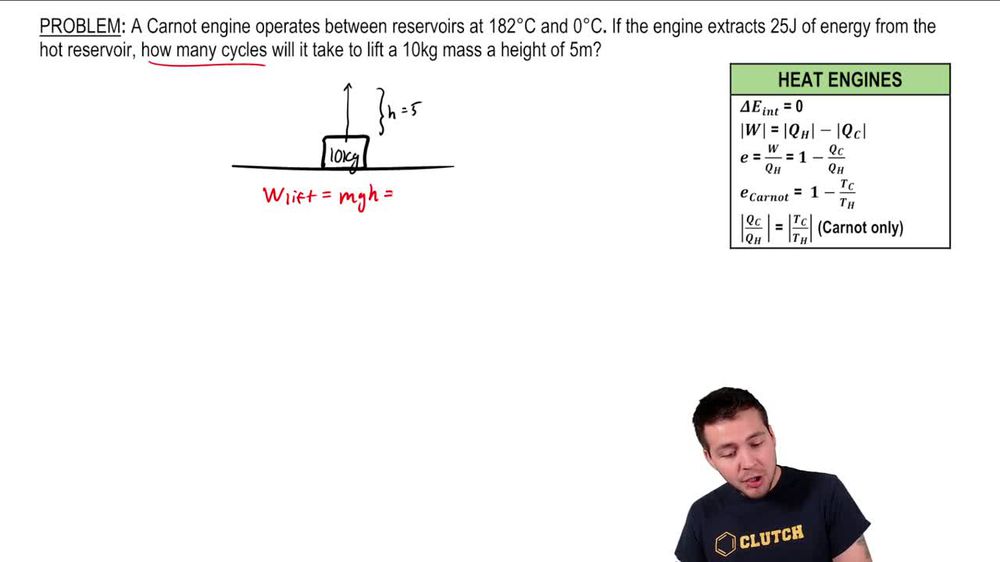
The Carnot Cycle
- Multiple ChoiceWhat is the Carnot efficiency of a heat engine operating between a hot reservoir at 300°C and a cold reservoir at 10°C?1351views
- Multiple Choice
A theoretical heat engine in space could operate between the Sun's 5500°C surface and the –270.3°C temperature of intergalactic space. What would be its maximum theoretical efficiency?
1388views6rank1comments - Textbook Question
(III) One mole of monatomic gas undergoes a Carnot cycle with TH = 350°C and TL = 210°C. The initial pressure is 8.8 atm. During the isothermal expansion, the volume doubles. Determine Q, W, and ∆Eint for each segment of the cycle.
864views - Textbook Question
One mole of monatomic gas undergoes a Carnot cycle with TH = 350°C and TL = 210°C. The initial pressure is 8.8 atm. During the isothermal expansion, the volume doubles. Calculate the efficiency of the cycle using Eqs. 20–1 and 20–3.
886views - Textbook Question
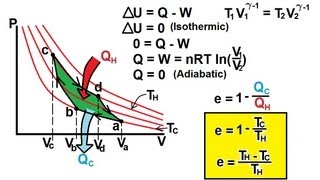
One mole of monatomic gas undergoes a Carnot cycle with TH = 350°C and TL = 210°C. The initial pressure is 8.8 atm. During the isothermal expansion, the volume doubles. Find the values of the pressure and volume at the points a, b, c, and d of Fig. 20–5.
900views - Textbook Question
A Carnot engine is operated between two heat reservoirs at temperatures of K and K. What is the thermal efficiency of the engine?
1291views - Textbook Question
(a) Show that the work done by a Carnot engine is equal to the area enclosed by the Carnot cycle on a PV diagram, Fig. 20–5. (See Section 19–7.) (b) Generalize this to any reversible cycle.
1007views - Textbook Question
Which, if any, of the heat engines in FIGURE EX21.22 violate (a) the first law of thermodynamics or (b) the second law of thermodynamics? Explain.
1229views - Multiple Choice
Your friend claims they have a design for a reversible heat engine that can operate between the freezing and boiling temperatures of water that has an efficiency of 30%. Is this possible?
1227views3rank - Textbook Question
The working substance of a certain Carnot engine is 1.0 mol of an ideal monatomic gas. During the isothermal expansion portion of this engine’s cycle, the volume of the gas doubles, while during the adiabatic expansion the volume increases by a factor of 6.2. The work output of the engine is 920 J in each cycle. Compute the temperatures of the two reservoirs between which this engine operates.
1473views - Textbook Question
A Carnot engine is operated between two heat reservoirs at temperatures of K and K. How much mechanical work is performed by the engine during each cycle?
1824views