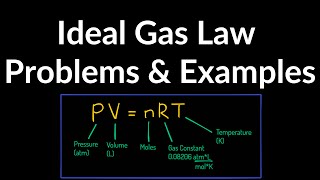
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
3 moles of an ideal gas fill a cubical box with a side length of 30cm. If the temperature of the gas is 20°C, what is the pressure inside the container?
1818views7rank - Multiple Choice
Hydrogen gas behaves very much like an ideal gas. If you have a sample of Hydrogen gas with a volume of 1000 cm3 at 30°C with a pressure of 1 × 105 Pa, calculate how many hydrogen atoms (particles) there are in the sample.
2805views8rank1comments - Multiple Choice
A balloon contains 3900cm3 of a gas at a pressure of 101 kPa and a temperature of –9°C. If the balloon is warmed such that the temperature rises to 28°C, what volume will the gas occupy? Assume the pressure remains constant.
1703views6rank - Multiple ChoiceA sample of gas is heated in a closed container with a volume of from an initial temperature and pressure of and to a final temperature of . What is the final pressure?1487views