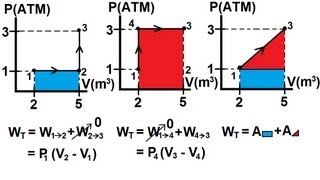
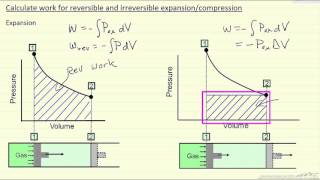
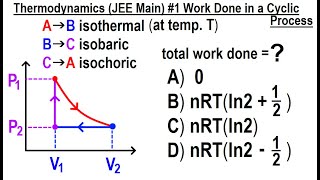
22. The First Law of Thermodynamics
Work Done Through Multiple Processes
22. The First Law of Thermodynamics
Work Done Through Multiple Processes
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
How much work is done on a gas that expands from A to B along the path shown below?
1374views3rank2comments - Multiple Choice
A gas with an initial volume of 0.2 m3 is heated at constant volume, and the pressure increases from 2×105 Pa to 5×105. Then, it compresses at constant pressure until it reaches a final volume of 0.12 m3. Draw the two processes in the PV diagram below and find the total work done by the gas.
1241views9rank - Multiple ChoiceHow much work is done on the gas in the process shown in the figure? Let , , , and .1565views
- Multiple ChoiceA proton with an initial speed of 750,000 m/s is brought to rest by an electric field. What was the potential difference that stopped the proton, given that the mass of a proton is approximately 1.67 x 10^-27 kg and its charge is 1.6 x 10^-19 C?608views