23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
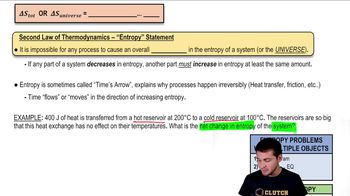
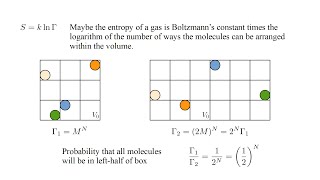
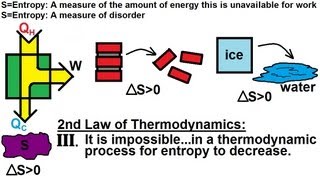
23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
3 moles of an ideal gas are compressed isothermally at 20°C. During this compression, 1850 J of work is done on the gas. What is the change of entropy of the gas?
2139views2rank1comments - Multiple Choice
You have a block of ice at 0°C. Heat is added to the ice, causing an increase in entropy of 120J/K. How much ice melts into water in this process?
1750views5rank - Multiple Choice
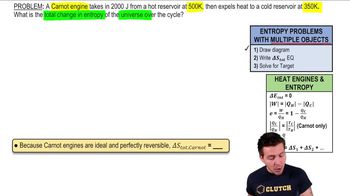
A non-Carnot heat engine operates between a hot reservoir at 610K and a cold reservoir at 320K. In a cycle, it takes in 6400 J of heat and does 2200 J of work. What is the total change in entropy of the universe over the cycle?
1716views2rank - Multiple Choice
Which of the following statements is consistent with the Second Law of Thermodynamics?
1253views