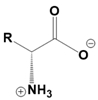
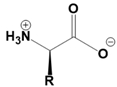
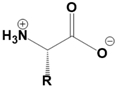
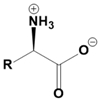
Amino acids are fundamental building blocks of proteins, and understanding their configuration is crucial in biochemistry. Most alpha amino acids are chiral, meaning they have non-superimposable mirror images, with glycine being the only achiral amino acid. When discussing amino acid chirality, biochemists often use Fisher's convention, which designates configurations as L and D instead of R and S. Notably, life predominantly utilizes L amino acids for protein synthesis across all domains of life, although D amino acids do have specific roles in certain bacteria.
All L amino acids typically possess an S configuration, with the exception of L cysteine, which has an R configuration due to the unique priority of its R group. To determine the configuration of amino acids, two methods can be employed: standard and non-standard Fisher projections. In standard Fisher projections, the carboxyl group is positioned at the top, the longest carbon chain is vertical, and the amino group is placed on the left for L amino acids. Conversely, for D amino acids, the amino group is not on the left.
In non-standard Fisher projections, the carboxyl group may not be at the top, and the longest carbon chain may not be vertical. The first step in analyzing these projections is to assign priorities to the groups around the chiral carbon. The nitrogen atom typically receives the highest priority, while the hydrogen atom has the lowest. For most amino acids, the R group is assigned priority three, but for cysteine, it is priority two. This distinction is crucial when determining the configuration.
When determining the configuration, if the arrangement of priorities appears clockwise, it indicates an R configuration, while a counterclockwise arrangement suggests an S configuration. However, if the fourth priority group is on a wedge, the configuration must be flipped. For example, if a non-standard projection of alanine shows a clockwise arrangement but the fourth priority is on a wedge, it is actually an S configuration, confirming it as L alanine. In contrast, cysteine, which has an R configuration, is identified as L cysteine.
Understanding these configurations and the methods to determine them is essential for further studies in biochemistry, particularly in protein structure and function. As you continue your studies, practice applying these concepts to various amino acids to solidify your understanding.