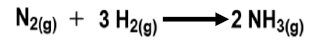Entropy is a fundamental concept in thermodynamics, often described as a measure of randomness or disorder within a system. Understanding entropy requires a grasp of the laws of thermodynamics, which govern the flow and transformation of heat, energy, and matter in both chemical reactions and biological processes.
The sun serves as the primary energy source for most living organisms on Earth. Through photosynthesis, plants and other photosynthetic organisms convert solar energy into chemical energy. This chemical energy is then transferred to animals when they consume plants, allowing for the conversion of this energy into mechanical or kinetic energy necessary for movement. It is important to note that during each energy transfer, some energy is inevitably lost as heat, a concept that will be explored further in relation to the second law of thermodynamics.
In thermodynamics, distinguishing between the system and its surroundings is crucial. The system refers to the specific part of the universe being studied, while the surroundings encompass everything else. Biological systems, such as living organisms, are classified as open systems because they can exchange both mass and energy with their surroundings. For instance, a cell is an open system defined by its plasma membrane, which allows for the import and export of substances and energy.
Additionally, specific chemical reactions can also be considered systems. For example, the conversion of glucose to glucose 6-phosphate within cells involves enzymes and essential ions like magnesium, along with ATP, which provides the necessary energy for the reaction. This interplay between biological systems and chemical reactions will be further examined in the context of entropy in future discussions.