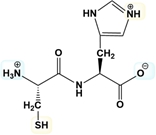
The isoelectric point (pI) of a peptide is a crucial concept in biochemistry, representing the pH at which the peptide has a net charge of zero. To calculate the pI, one must focus on the pKa values associated with the ionizable groups of the amino acids within the peptide. The pI is determined by averaging the two pKa values that flank the pH at which the predominant structure of the peptide is neutral.
To begin, it is essential to understand the ionization behavior of the amino acid side chains. There are seven amino acids with ionizable R groups, which can be remembered using the mnemonic "yucky crazy dragons knights riding horses." Knowing these amino acids is the first step in calculating the pI.
The calculation process involves four key steps:
Identify the ionizable groups in the peptide. This includes the amino group at the N-terminus, the carboxyl group at the C-terminus, and any ionizable side chains of the amino acids.
Order the pKa values of these groups from smallest to largest. This ordering is crucial as it helps in determining the pH ranges for subsequent calculations.
Determine the net charge of the predominant structures at various pH levels between the ordered pKa values. This involves comparing the pH to each pKa: if the pH is lower than the pKa, the protonated (conjugate acid) form predominates; if higher, the deprotonated (conjugate base) form predominates.
Calculate the pI by averaging the two pKa values that correspond to the pH range where the net charge is zero. This is the pH at which the peptide is electrically neutral.
For example, consider the peptide composed of aspartic acid (D), glycine (G), and glutamic acid (E). The relevant pKa values for the ionizable groups are as follows: the carboxyl group of the C-terminus (pKa = 3.5), the R group of aspartic acid (pKa = 3.9), the R group of glutamic acid (pKa = 4.1), and the amino group at the N-terminus (pKa = 8.0). By ordering these values and determining the net charge at various pH levels, one can find the pH range where the net charge is zero.
In this case, if the pH is chosen between 3.5 and 3.9, the net charge can be calculated, leading to the identification of the pI. The final calculation involves averaging the two pKa values that sandwich the neutral charge, yielding the pI of the peptide.
Understanding the isoelectric point is vital for predicting the behavior of peptides in different pH environments, which has implications in protein purification, formulation, and stability studies.