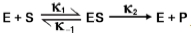Michaelis and Menten are pivotal figures in the study of enzyme kinetics, having introduced a fundamental model in 1913 that significantly advanced our understanding of how enzymes function. Leonor Michaelis, a German scientist, and Maud Menten, a Canadian woman, proposed that for an enzyme-catalyzed reaction to occur, an enzyme-substrate complex must first form. This was a departure from earlier beliefs that enzymes merely accelerated reactions by releasing some chemical agent.
Their model laid the groundwork for what is now known as Michaelis-Menten kinetics, which describes the rate of enzymatic reactions. The Michaelis-Menten equation, although not the focus of this discussion, is essential for quantifying enzyme activity and will be explored in detail later. It is important to note that this equation operates under specific assumptions that must be met for accurate application.
Maud Menten's contributions are particularly remarkable given the societal challenges she faced as a woman in the early 20th century, a time when Canadian law did not recognize women as persons. Her achievements as a medical doctor and a PhD holder underscore her significant role in the field of enzymology.
As we progress in this course, we will delve deeper into the Michaelis-Menten equation and the assumptions necessary for its application, enhancing our understanding of enzyme kinetics and their implications in biological systems.