9. Quantum Mechanics
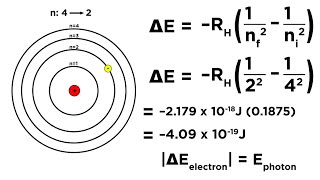
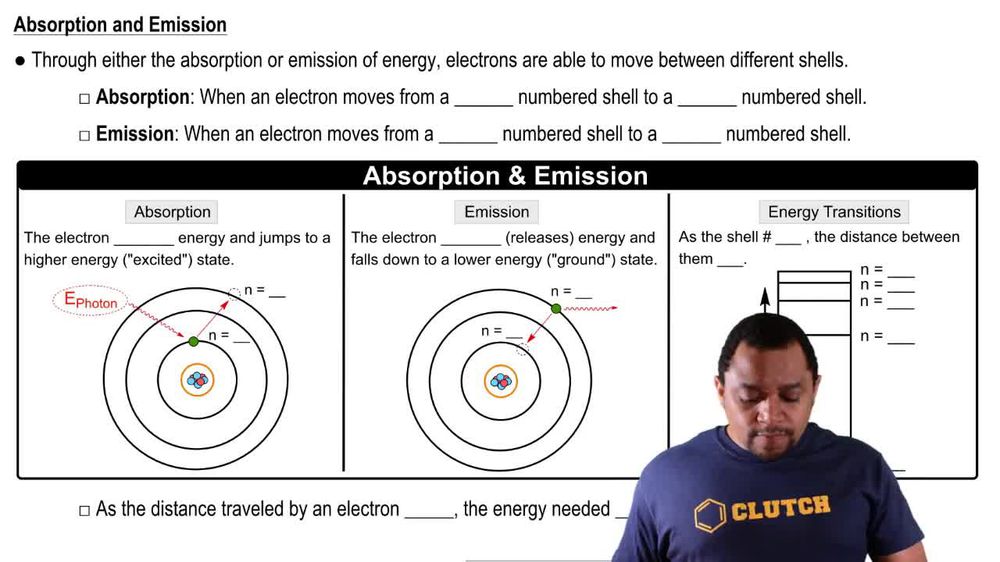
Bohr Model
Practice this topic
- Multiple Choice
Which of the electron transitions represents absorption with the greatest frequency?
3672views12rank - Multiple Choice
Which of the following transitions (in a hydrogen atom) represent emission of the shortest wavelength?
5961views6rank2comments - Multiple Choice
If the energy of an electron within the boron atom was calculated as –6.0556 x 10-18 J, at what energy level would it reside?
1936views4rank3comments - Multiple ChoiceWhich of the following statements about the Bohr model of the hydrogen atom is false?1043views
- Textbook QuestionWhich arrow in the energy diagram for an atom represents the absorption of light with the shortest wavelength? (LO 5.7)1279views1rank
- Textbook Question
Consider the three electronic transitions in a hydrogen atom shown here, labeled A, B, and C. (a) Three electromagnetic waves, all drawn on the same scale, are also shown. Each corresponds to one of the transitions. Which electromagnetic wave (i), (ii), or (iii), is associated with electronic transition C?
1397views2rank - Textbook Question
Does the hydrogen atom 'expand' or 'contract' when an electron is excited from the n = 1 state to the n = 3 state?
1095views - Textbook Question
Classify each of the following statements as either true or false: (a) A hydrogen atom in the n = 3 state can emit light at only two specific wavelengths (b) a hydrogen atom in the n = 2 state is at a lower energy than one in the n = 1 state (c) the energy of an emitted photon equals the energy difference of the two states involved in the emission.
662views