13. Liquids, Solids & Intermolecular Forces
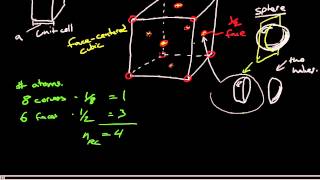
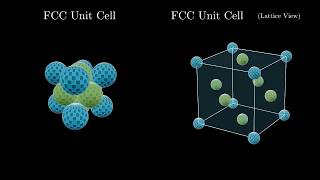
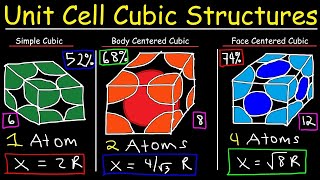
Face Centered Cubic Unit Cell
13. Liquids, Solids & Intermolecular Forces
Face Centered Cubic Unit Cell
Additional 2 creators.
Learn with other creators
Showing 5 of 5 videos
Practice this topic
- Multiple Choice
Aluminum has a face-centered cubic unit structure and a density of 2.716 g/cm3. Calculate the edge length of the unit cell.
2672views6rank2comments - Multiple Choice
An element crystallizes in a face-centered cubic lattice and has a density of 3.16 g/cm3. The edge of its unit cell is 7.49 x 10-8 cm. How many atoms are in each unit cell?
1511views2rank - Multiple Choice
An element crystallizes in a face-centered cubic lattice and has a density of 18.44 g/cm3. The edge of its unit cell is 1.05×10-8 cm. Calculate the atomic mass for the element.
1607views1rank - Multiple Choice
Sodium chloride solidifies into a face-centered cubic unit cell with a density of 2.17 g/cm3. What is its volume in cm3?
1448views1rank - Open Question
Assuming that the ionic radius of oxygen is 140 pm, estimate the ionic radius of manganese.
888views