6. Chemical Quantities & Aqueous Reactions
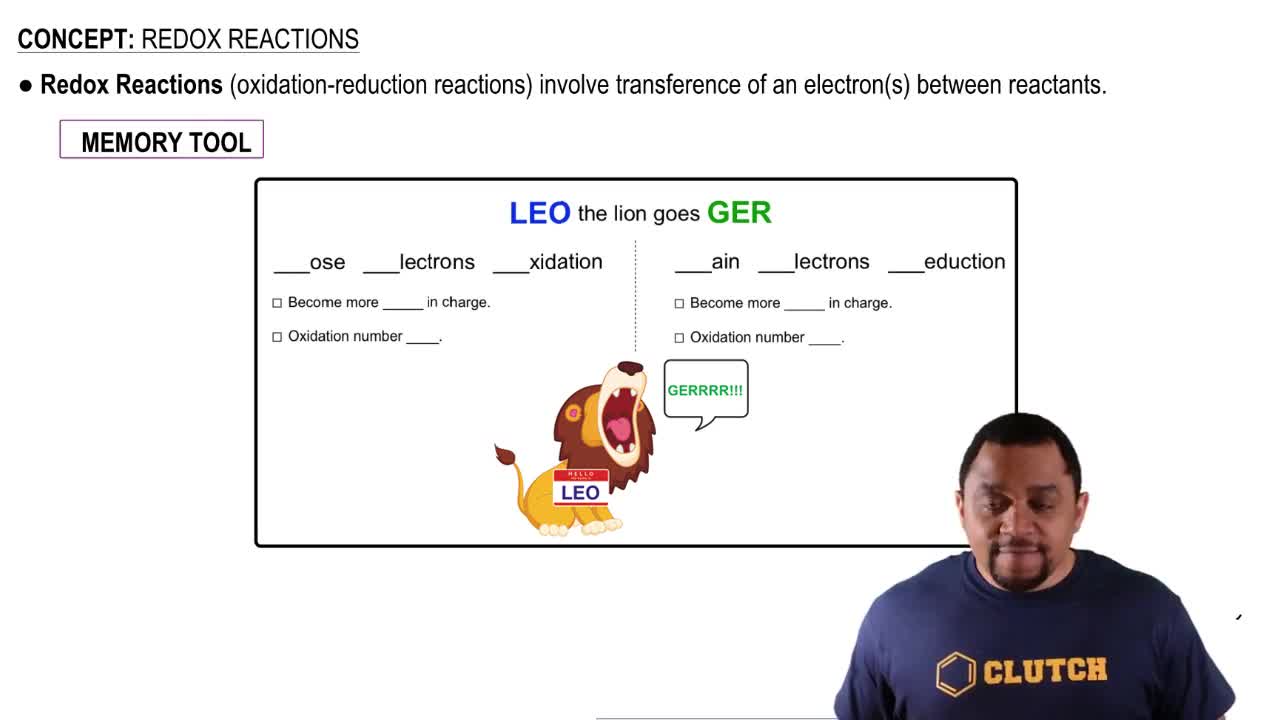
Redox Reactions
Learn with other creators
Practice this topic
- Multiple Choice
Which element is being reduced in the following reaction?
Cr2O72- + 3 HNO2 + 5 H+ → 2 Cr3+ + 3NO3- + 4 H2O
4876views5rank3comments - Multiple Choice
Identify the oxidizing agent and reducing agent from the following redox reaction.
Ba (s) + Cl2 (g) → BaCl2 (aq)
6033views11rank1comments - Multiple Choice
Which element is oxidized and which is reduced in the following reaction?
Hg (aq) + HgCl2 (aq) → Hg2Cl2
4736views9rank1comments - Multiple Choice
Which of the following represents an oxidation-reduction reaction?
I. PCl3 (aq) + Cl2 (g) → PCl5 (aq)
II. 2 AgNO3 (aq) + Cu (s) → Cu(NO3)2 (aq) + 2 Ag (s)
III. CO2 (g) + 2 LiOH (aq) → Li2CO3 (aq) + H2O (l)
IV. FeCl2 (aq) + 2 NaOH (aq) → Fe(OH)2 (aq) + 2 NaCl (aq)
4692views17rank4comments - Open Question
A substance with the potential to cause reduction in another substance is __________.
997views - Open Question
A substance with the potential to cause oxidation in another substance is __________.
1006views - Open Question
Which of the following is true about a redox reaction? In a redox reaction, the reducing agent loses electrons. In a redox reaction, the oxidizing agent loses electrons. In a redox reaction, the oxidized species gains electrons. In a redox reaction, the reduced species loses electrons.
1011views - Open Question
Identify the oxidizing agent and the reducing agent for 4Li(s) + O2(g) → 2Li2O(s)
1074views