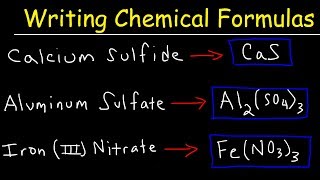
3. Chemical Reactions
Writing Ionic Compounds
3. Chemical Reactions
Writing Ionic Compounds
Additional 6 creators.
Learn with other creators
Showing 9 of 9 videos
Practice this topic
- Multiple Choice
Provide the molecular formula for the following compound:Copper (I) nitrate
2389views8rank2comments - Multiple Choice
Provide the molecular formula for the following compound:Sodium dichromate
5348views25rank2comments - Multiple ChoiceWhich of the following statements is false?1208views
- Open Question
Predict the chemical formula for the ionic compound formed by the elements Ca and Br.
1056views - Open QuestionWhen calcium reacts with sulfur the compound formed is1589views
- Open Question
When magnesium (Mg2+) and fluorine (F1-) combine in an ionic bond, the resulting formula will be
1382views - Open Question
What is the formula of a compound that contains Na+ and PO43- ions?
1184views - Multiple ChoiceNa reacts with element X to form an ionic compound with the formula Na₃X. Based on this information, what is the likely formula for the ionic compound formed between Al and X?701views