9. Quantum Mechanics
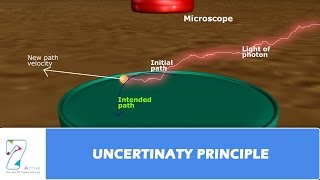
Heisenberg Uncertainty Principle
9. Quantum Mechanics
Heisenberg Uncertainty Principle
Additional 5 creators.
Learn with other creators
Showing 8 of 8 videos
Practice this topic
- Multiple Choice
To what uncertainty (in m) can the position of a baseball traveling at 51.0 m/s be measured if the uncertainty of its speed is 0.12%? The mass of the baseball is 150 g.
1730views4rank3comments - Multiple Choice
A proton with a mass of 1.67 x 10-27 kg traveling at 4.7 x 105 m/s has an uncertainty in its velocity of 1.77 x 105 m/s. Determine its uncertainty in position.
2002views3rank4comments - Multiple ChoiceWhich of the following statements about the Heisenberg uncertainty principle is false?1660views
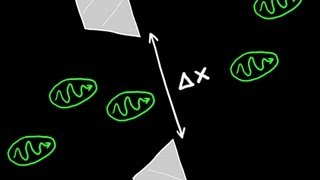
- Open QuestionWhat is the uncertainty of the position of the bacterium?1243views
- Multiple Choice
An electron with a mass of 9.11 x 10-31 kg has an uncertainty in its position of 630 pm. What is the uncertainty in its velocity?
1003views8rank