7. Gases
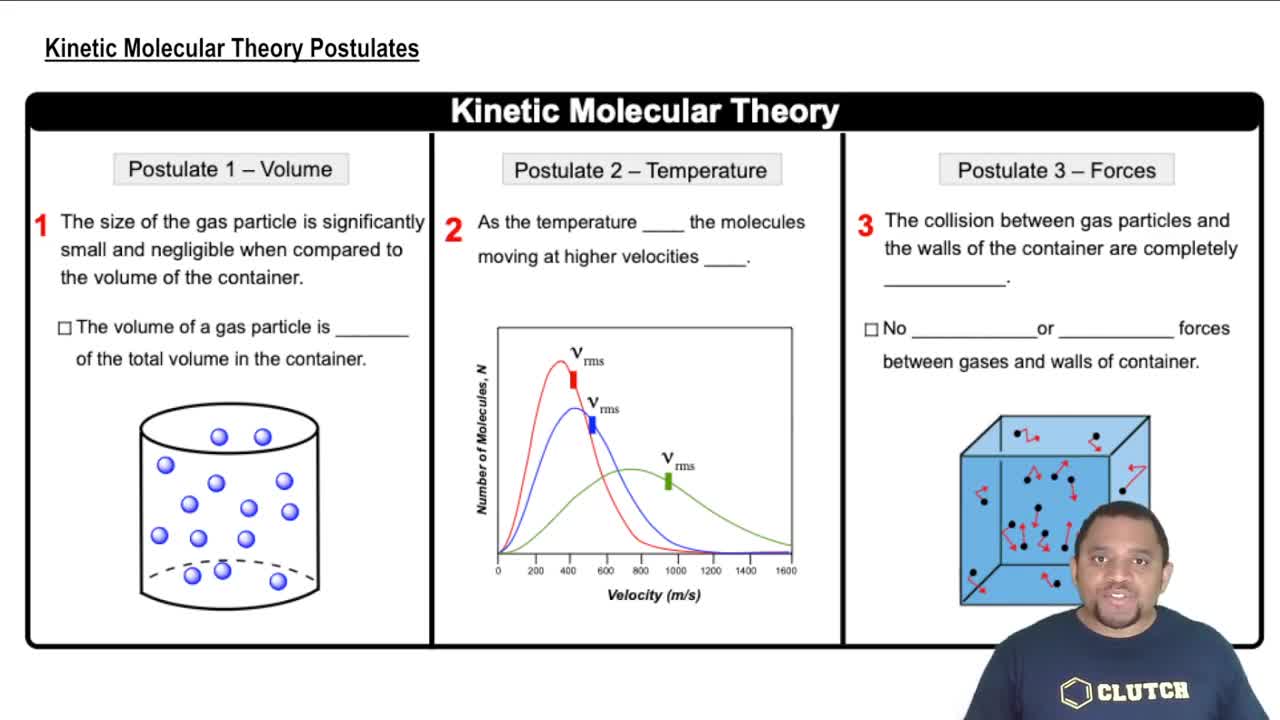
Kinetic Molecular Theory
Learn with other creators
Practice this topic
- Multiple Choice
Which of the following statements would correctly explain the non-ideal behavior of a gas based on the Kinetic Molecular Theory (KMT)?
a) At high temperatures the attractive forces between molecules becomes negligible.
b) At high pressure the volume of gas molecules become significant.
c) An increase or decrease in the moles of gas causes the gas constant value to change.3726views9rank - Multiple Choice
Which of the following statements is/are true for gas molecules according to the Kinetic Molecular Theory?
I.Increasing the amount of gas molecules increases the pressure by increasing the force of the collisions.
II.Decreasing the temperature of a gas decreases the pressure by increasing the force of the collisions.
III.Decreasing the volume of a gas increases pressure by increasing the frequency of the collisions.4628views9rank - Multiple Choice
Based on the kinetic-molecular theory, which of the following is/are true?
I.At a given temperature, all gases have the same average kinetic energy.
II.At a given temperature, different gases have the same average velocities.
III.The average kinetic energy is proportional to the absolute temperature.3731views15rank2comments - Multiple Choice
Which statement is TRUE about kinetic molecular theory?
a) A single particle does not move in a straight line.
b) The size of the particle is large compared to the volume.
c) The collisions of particles with one another is completely elastic.
d) The average kinetic energy of a particle is not proportional to the temperature.4315views6rank - Open Question
Which temperature represents the highest average kinetic energy?
911views - Open QuestionWhich of the gases has the fastest‑moving molecules, on average, at a given temperature?985views