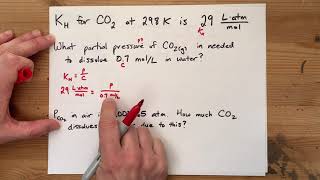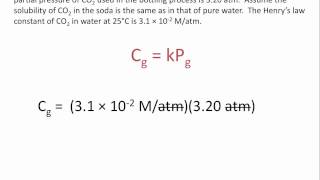14. Solutions
Henry's Law Calculations
Learn with other creators
Practice this topic
- Multiple Choice
Henry's Law Constant for nitrogen in water is 1.67 × 10-4 M • atm–1. If a closed canister contains 0.103 M nitrogen, what would be its pressure in atm?
3558views8rank1comments - Multiple Choice
At 0°C and 1.00 atm, as much as 0.84 g of O2 can dissolve in 1.0 L of water. At 0°C and 4.00 atm, how many grams of O2 dissolve in 1.0 L of water?
2706views8rank - Multiple Choice
The atmospheric pressure in a lab is calculated as 1.3 atm. If oxygen gas contributes 62% of this atmospheric pressure, determine its mass (in g) dissolved at room temperature in 25 L of water. The Henry's Law Constant for oxygen in water at this temperature is 5.3 × 10–5 M/atm.
2492views11rank - Multiple ChoiceWhat would be the vapor pressure at 25.0 °C of a solution of 5.00 g of glucose (C6H12O6) in 100.0 g of ethanol (C2H5OH)? Pure ethanol has a vapor pressure of 54.68 mmHg at 25 °C.1417views
- Open Question
At 298 K, the Henry's law constant for oxygen is 0.00130 M/atm. Air is 21.0% oxygen.
1662views - Open Question
At 298 k, the Henry's law constant for oxygen is 0.00130 m/atm. Air is 21.0% oxygen. at 298 k, what is the solubility of oxygen in water exposed to air at 1.00 atm?
1109views - Open Question
At 298 K, the Henry's law constant for oxygen is 0.00130 M/atm. Air is 21.0% oxygen. At 298 K, what is the solubility of oxygen in water exposed to air at 1.00 atm?
1806views