9. Quantum Mechanics
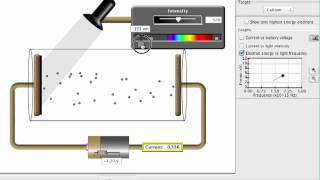
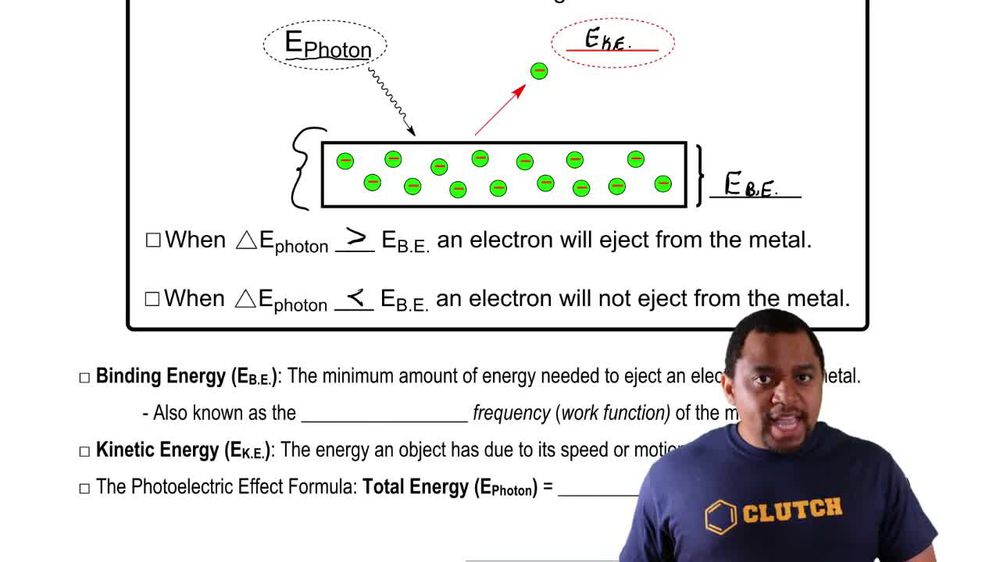
Photoelectric Effect
Practice this topic
- Multiple Choice
A metal with a threshold frequency of 2.15 x 1015 s-1 emits an electron with a velocity of 7.03 x 106 m/s when radiation of 4.88 x 1015 s-1 strikes the metal's surface. Calculate the mass of the electron.
4090views10comments - Multiple Choice
An ultraviolet photon with a wavelength of 320 nm strikes a metal surface. The emitted electron has a kinetic energy of 3.0 x 10-2 eV. What is the binding energy of the electron in kJ/mol? 1 electron volt (eV) = 1.602 x 10-19 J.
2387views5rank2comments - Multiple ChoiceWhich best describes the photoelectric effect?1230views
- Multiple ChoiceWhich of the following is true about the photoelectric effect?1281views
- Textbook QuestionWhich type of electromagnetic radiation will cause the greatest number of electrons to be ejected from zinc metal with a work function of 350 kJ/mol? (LO 5.4, 5.5)(a) Dim light with a wavelength of 320 nm (b) Dim light with a wavelength of 360 nm (c) Bright light with a wavelength of 360 nm (d) Bright light with a wavelength of 375 nm806views1rank
- Textbook QuestionEinstein's 1905 paper on the photoelectric effect was thefirst important application of Planck's quantum hypothesis.Describe Planck's original hypothesis, and explain howEinstein made use of it in his theory of the photoelectriceffect.1139views
- Textbook Question
Molybdenum metal must absorb radiation with an energy higher than 7.22 * 10-19 J ('energy threshold') before it can eject an electron from its surface via the photoelectric effect. (c) If molybdenum is irradiated with light of wavelength of 240 nm, what is the maximum possible velocity of the emitted electrons?
637views1comments - Textbook Question
Molybdenum metal must absorb radiation with an energy higher than 7.22 * 10-19 J ('energy threshold') before it can eject an electron from its surface via the photoelectric effect. (b) What wavelength of radiation will provide a photon of this energy?
655views1comments