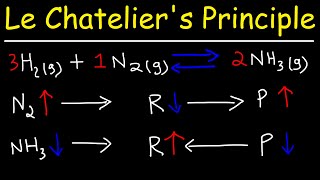
16. Chemical Equilibrium
Le Chatelier's Principle
Learn with other creators
Practice this topic
- Multiple Choice
Consider the reaction below:
CH4 (g) + F2 (g) ⇌ CF4 (g) + HF (g) H = + 38.1 KJ/mol
The following changes will shift the equilibrium to the left except one. Which one would not cause a shift to the left?
3281views6rank8comments - Multiple Choice
The following data was collected for the following reaction at equilibrium
2 A (g) + 3 B (g) ⇌ C (g)
At 25 oC K is 5.2 x 10-4 and at 50 oC K is 1.7 x 10-7. Which of the following statements is true?
a) The reaction is exothermic.
b) The reaction is endothermic.
c) The enthalpy change, ΔH, is equal to zero.
d) Not enough information is given.
2393views4rank6comments - Multiple Choice
Which direction will the following reaction (in a 10.0 L flask) proceed if a catalyst is added to the system?
CaCO3 (s) ⇌ CaO (s) + CO2 (g) Kp = 3.2 x 10-28
2878views6rank1comments - Multiple Choice
Consider the following gas reaction of A2 ( shaded spheres) and B2 ( unshaded spheres)
A2 (g) + B2 (g) ⇌ 2 AB (g)
Which container proceeds more to completion?
2864views7rank4comments