18. Aqueous Equilibrium
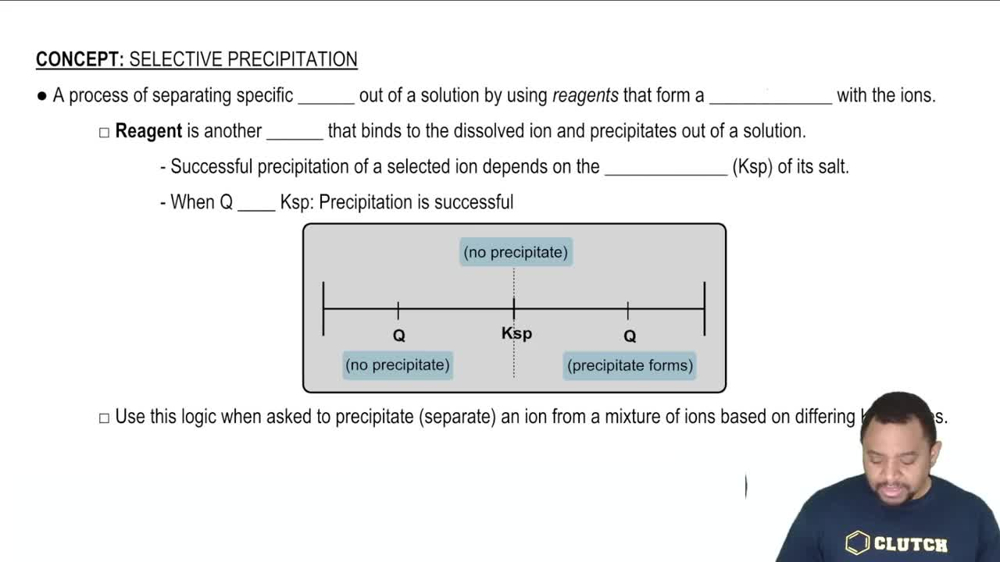
Selective Precipitation
Practice this topic
- Multiple Choice
Solution contains [Cu2+] = 0.035 M, [Sr2+] = 0.054 M, [Al3+] = 0.23 M. Cu2+ can be separated by selective precipitation using NaOH. What is the minimum concentration of NaOH needed to start precipitation of Cu2+? (Ksp = 2.2 × 10−20 of Cu(OH)2).
621views4rank - Textbook Question
Suppose that a 10-mL sample of a solution is to be tested for I- ion by addition of 1 drop (0.2 mL) of 0.10 M Pb1NO322. What is the minimum number of grams of I- that must be present for PbI21s2 to form?
506views1comments - Textbook Question
The water supply for a midwestern city contains the following impurities: coarse sand, finely divided particulates, nitrate ions, trihalomethanes, dissolved phosphorus in the form of phosphates, potentially harmful bacterial strains, dissolved organic substances. Which of the following processes or agents, if any, is effective in removing each of these impurities: coarse sand filtration, activated carbon filtration, aeration, ozonization, precipitation with aluminum hydroxide?
406views - Textbook Question
(b) What is the most significant difference between the sulfides precipitated in group 2 and those precipitated in group 3?
465views - Textbook Question
Determine the minimum concentration of the precipitating agent on the right to cause precipitation of the cation from the solution on the left. c. 0.0018 M AgNO3; RbCl
513views