18. Aqueous Equilibrium
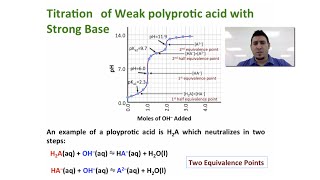
Titrations: Diprotic & Polyprotic Buffers
18. Aqueous Equilibrium
Titrations: Diprotic & Polyprotic Buffers
Additional 1 creators.
Learn with other creators
Practice this topic
- Multiple Choice
Calculate the pH of 75.0 mL of a 0.10 M of phosphorous acid, H3PO3, when 80.0 mL of 0.15 M NaOH are added. Ka1 = 5.0 × 10−2, Ka2 = 2.0 × 10−7.
1070views2rank2comments - Multiple Choice
Find the pH when 100.0 mL of a 0.1 M dibasic compound B (pKb1 = 4.00; pKb2 = 8.00) was titrated with 11 mL of a 1.00 M HCl.
1026views2rank1comments - Multiple Choice
Suppose you have 50.1 mL of a H3PO4 solution that you titrate with 15.4 mL of 0.10 M KOH solution to reach the endpoint. What is the concentration of H3PO4 of the original H3PO4 solution?
1010views1rank1comments - Open QuestionA 25.0 ml sample of 0.125 moll−1 pyridine (kb=1.7×10−9) is titrated with 0.100 moll−1hcl.866views
- Open Question
What is observed when equal volumes of 0.1 M aqueous HCl and 0.01 M aqueous Na2SO3 are mixed?
1033views - Open Question
What is the identity of the acid (H3X) identified via titration of Na3X with HCl?
1191views - Multiple ChoiceThe titration of 18.0 mL of an unknown diprotic acid requires 15.3 mL of 0.120 M KOH solution to reach the equivalence point. What is the concentration of the unknown acid sample?697views