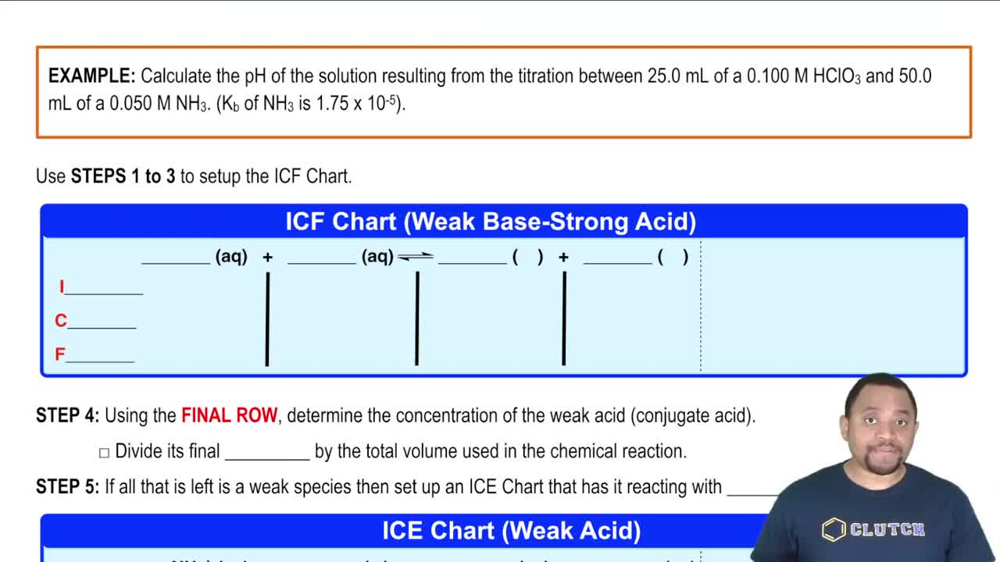
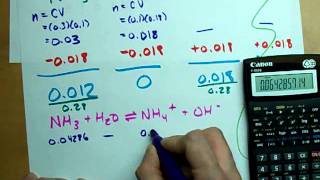18. Aqueous Equilibrium
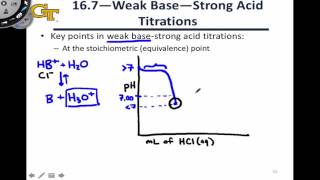
Titrations: Weak Base-Strong Acid
Learn with other creators
Practice this topic
- Multiple ChoiceWhat is the initial pH for the titration of 30.0 mL of 0.850 M NH3 with 0.100 M HCl. Kb for NH3 = 1.8 × 10−5.704views
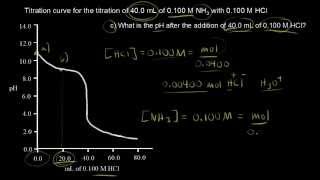
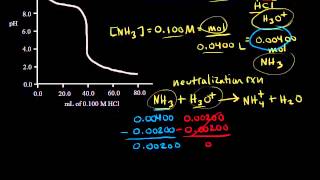
- Multiple ChoiceThe plot below illustrates which type of titration?743views
- Multiple Choice
Calculate the pH of the solution resulting from the mixing of 75.0 mL of 0.100 M NaC2H3O2 and 75.0 mL of 0.30 M HC2H3O2 with 0.0040 moles of HBr.
765views1rank2comments - Multiple Choice
In order to create a buffer 7.321 g of potassium lactate is mix with 550.0 mL of 0.328 M lactic acid, HC3H5O3. What is the pH of the buffer solution after the addition of 300.0 mL of 0.100 M hydrobromic acid, HBr? The Ka of HC3H5O3 is 1.4 × 10−4.
617views4rank - Textbook Question
Assume that 30.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a 0.10 M solution of the monoprotic strong acid HA. (b) What is the predominant form of B at the equivalence point?
894views - Textbook Question
Assume that 30.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a 0.10 M solution of the monoprotic strong acid HA. (a) How many moles of HA have been added at the equivalence point?
618views - Textbook Question
The following pictures represent solutions at various stages in the titration of a weak base B with aqueous HCl. (Cl- ions and solvent water molecules have been omitted for clarity.)
. (a) To which of the following stages do solutions 1–4 correspond? (i) The initial solution before addition of any HCl (ii) Halfway to the equivalence point (iii) At the equivalence point (iv) Beyond the equivalence point
809views - Textbook Question
How many milliliters of 0.105 M HCl are needed to titrate each of the following solutions to the equivalence point: (b) 22.5 mL of 0.118 M NH3?
1022views