1. Intro to General Chemistry
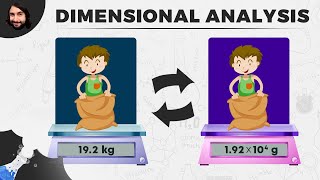
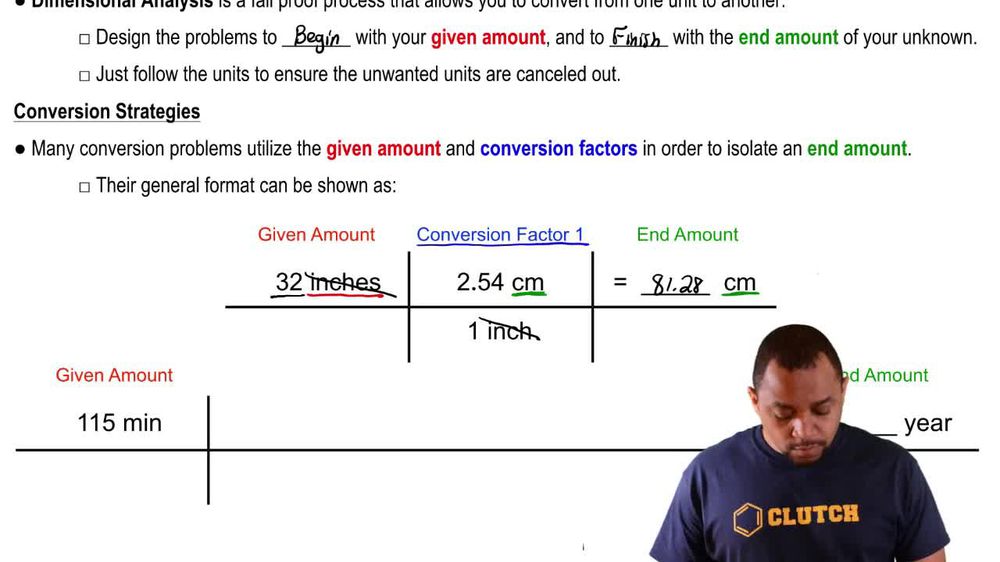
Dimensional Analysis
1. Intro to General Chemistry
Dimensional Analysis
Showing 9 of 9 videos
Practice this topic
- Multiple Choice
If the distance between Washington, D.C. and New York City is 224.9 miles, determine the distance in centimeters.
4038views17rank5comments - Multiple Choice
Examine the following graph.
Which of the following is correct?
1234views - Multiple Choice
A large scale ranching farm possesses 22.5 million acres with 5.0 x 10-5 % reserved for housing. Determine how many square kilometers this area for dwelling represents. (1 km = 0.6214 miles) (1 mile = 5280 ft) (1 acre = 43,560 ft2)
1181views16rank1comments - Textbook QuestionConsider the jar of jelly beans in the photo. To get an estimateof the number of beans in the jar you weigh six beansand obtain masses of 3.15, 3.12, 2.98, 3.14, 3.02, and 3.09 g.Then you weigh the jar with all the beans in it, and obtain amass of 2082 g. The empty jar has a mass of 653 g. Based onthese data, estimate the number of beans in the jar. Justifythe number of significant figures you use in your estimate.885views
- Textbook QuestionA person runs at a pace of 6.52 mi/hr. How long does it take the person to run a 15.0 km race? (1 mi = 1.61 km) (LO 1.17) (a) 85.7 min (b) 222 min(c) 50.0 min (d) 93.4 min535views
- Textbook Question
The radius of an atom of tungsten (W) is about 2.10 Å. (c) If the atom is assumed to be a sphere, what is the volume in m3 of a single W atom?
423views - Textbook Question
The radius of an atom of tungsten (W) is about 2.10 Å. (b) How many tungsten atoms would have to be lined up to create a wire of 2.0 mm?
809views - Multiple ChoiceWhich of the following steps is essential for performing dimensional analysis?532views