2. Atoms & Elements
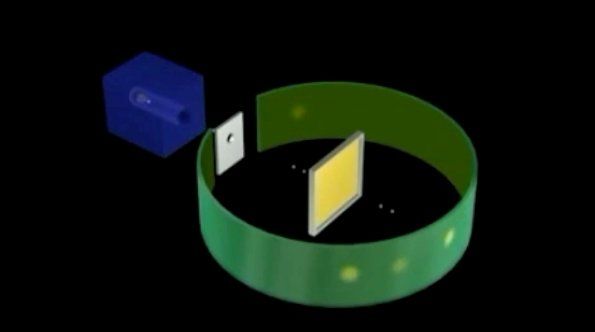
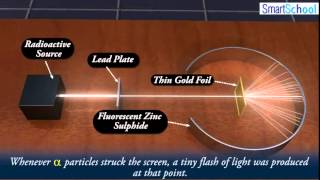
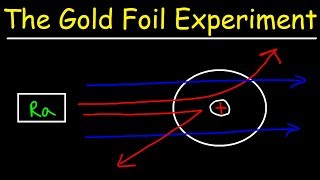
Rutherford Gold Foil Experiment
2. Atoms & Elements
Rutherford Gold Foil Experiment
Additional 11 creators.
Learn with other creators
Showing 14 of 14 videos
Practice this topic
- Multiple ChoiceIn the gold foil experiment,1149views
- Textbook Question
Bromine has two naturally occurring isotopes; 79Br (mass of 78.918) and 81Br (mass of 80.916). If the atomic weight of bromine is 79.904, predict the mass spectrum of a sample of bromine atoms. (LO 2.17) (a)
(b)
(c)
(d)
539views - Textbook QuestionDiscovering which of the three subatomic particles proved to bethe most difficult—the proton, neutron, or electron? Why?371views
- Textbook QuestionWhat fraction of the a particles in Rutherford's gold foil experimentare scattered at large angles? Assume the gold foilis two layers thick, as shown in Figure 2.9, and that the approximatediameters of a gold atom and its nucleus are 270pm and 1.0 * 10–2 pm, respectively. Hint: Calculate the crosssectional area occupied by the nucleus as a fraction of thatoccupied by the atom. Assume that the gold nuclei in eachlayer are offset from each other.1543views2rank
- Textbook Question
Consider the mass spectrometer shown in Figure 2.11. Determine whether each of the following statements is true or false. If false, correct the statement to make it true: (b) The height of each peak in the mass spectrum is inversely proportional to the mass of that isotope.
529views - Multiple ChoiceIn Francesco Redi's experiment, what did he discover about the origin of maggots on decaying meat?667views
- Multiple ChoiceWhat did Ernest Rutherford's gold foil experiment demonstrate about the structure of atoms?86views
- Multiple ChoiceWhen Ernest Rutherford directed a beam of alpha particles at a thin sheet of gold foil, what did he expect would happen to the majority of the particles?50views