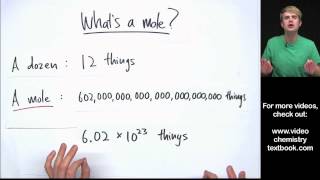2. Atoms & Elements
Mole Concept
Learn with other creators
Practice this topic
- Multiple Choice
If a sample of sodium chloride, NaCl, contains 73.1 kg, what is its number of formula units?
6328views23rank1comments - Multiple Choice
Calculate the number of oxygen atoms found in 783.9 g NiCl2 • 6 H2O.
5115views19rank3comments - Multiple Choice
If the density of water is 1.00 g/mL at 25°C, calculate the number of water molecules found in 1.50 x 103 µL of water.
5841views7rank3comments - Multiple Choice
A cylindrical copper wire is used for the fences around a house. The copper wire has a diameter of 0.0750 in. How many copper atoms are found in 5.160 cm piece? The density of copper is 8.96 g/cm3. (V = π • r2 • h)
5084views28rank - Open Question
How many atoms of oxygen are in the chemical formula 2Ca(ClO2)2? 2, 4, 6, or 8?
1462views - Open Question
How many moles are there in 8.50 x 10^24 molecules of sodium sulfate, Na2SO3?
1579views - Open Question
How many moles are in 2.5 g of N2? 0.089 moles, 0.18 moles, 1.3 moles, 11 moles
1577views - Open Question
What is the number of moles of CO2 in a 220 gram sample of CO2?
1357views