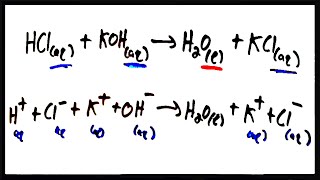6. Chemical Quantities & Aqueous Reactions
Complete Ionic Equations
Practice this topic
- Multiple Choice
Provide the net ionic equation that occurs when the following aqueous compounds are mixed together:
Copper (II) Bromide and Lithium Hydroxide
2471views13rank4comments - Multiple Choice
Which of the following reagents could be used to separate the two anions from a solution containing magnesium nitrate and cesium hydroxide?
2408views8rank - Multiple Choice
Which of the following reagents could be used to separate the two cations from a solution containing Lead (IV) acetate and cesium permanganate?
2483views9rank - Multiple ChoiceAn unknown sample is analyzed using the general qualitative analysis scheme shown in Figure 18.16. The sequence of reagents shown in the figure are used and a precipitate in only produced when (NH4)2HPO4 in NH3 is added. Which of the following describes a set of ions that might be present in the solution?816views
- Textbook Question
Which of the following ions will always be a spectator ion in a precipitation reaction? (a) Cl- (b) NO3- (c) NH4+ (d) S2- (e) SO42-
2322views - Textbook QuestionWrite a net ionic equation for the reaction that occurs when 10 mL of 0.5 M ammonium carbonate is mixed with 10 mL of 0.5 M silver nitrate. (LO 4.9, 4.11) (a) (b) (c) (d) A net ionic reaction cannot be written because a reaction does not take place.557views
- Textbook Question
Which ions remain in solution, unreacted, after each of the following pairs of solutions is mixed? (a) potassium carbonate and magnesium sulfate
646views - Textbook Question
Which ions remain in solution, unreacted, after each of the following pairs of solutions is mixed? (c) ammonium phosphate and calcium chloride
903views