11. Bonding & Molecular Structure
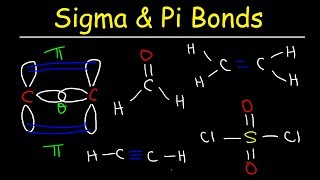
Lewis Dot Structures: Sigma & Pi Bonds
Practice this topic
- Multiple Choice
How many pi bonds does the following molecule contain?
2407views9rank - Multiple Choice
How many sigma bonds does the following molecule contain?
3318views9rank - Multiple Choice
Which has greater bond strength between the carbon–carbon bond.
C2Cl2 vs. C2Cl6
2296views10rank1comments - Multiple Choice
Draw the total number of sigma and pi bonds of the sulfur trioxide molecule, SO3.
2512views7rank5comments - Textbook QuestionWhich molecule has the shortest carbon–oxygen bond? (LO 7.3, 7.9) (a) CH3OH(b) CO (c) CO2 (d) HCOOH1312views1rank
- Textbook Question
The partial Lewis structure that follows is for a hydrocarbon molecule. In the full Lewis structure, each carbon atom satisfies the octet rule, and there are no unshared electron pairs in the molecule. The carbon—carbon bonds are labeled 1, 2, and 3. (c) Which carbon—carbon bond is the strongest one?
362views - Textbook Question
In the hydrocarbon (b) How many s bonds are there in the molecule?
447views - Textbook Question
The drawing below shows the overlap of two hybrid orbitals to form a bond in a hydrocarbon. (a) Which of the following types of bonds is being formed: (i) C¬C s, (ii) C¬C p, or (iii) C¬H s?
464views