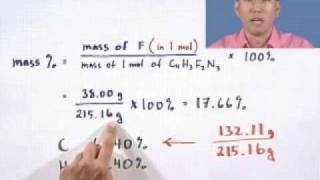3. Chemical Reactions
Mass Percent
3. Chemical Reactions
Mass Percent
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
Determine the percent composition of nitrogen and oxygen with nitrogen dioxide, NO2.
5436views33rank2comments - Multiple ChoiceA 70.0-g iron nail combines with 30.0 g of oxygen to form rust. What is the total mass of the iron nail plus the rust that forms?1518views
- Multiple ChoiceA nitrogen oxide is 63.65% by mass nitrogen. The molecular formula could be:2294views
- Multiple ChoiceThe following data were collected when analyzing a compound used in dentistry as a cement to repair teeth:
50.82% Zn;16.04% P;33.14% O
What is the empirical formula of the compound?1551views - Open Question
A compound whose empirical formula is XF3 consists of 65% F by mass. What is the atomic mass of X?
1334views - Open Question
What is the mass % of carbon in dimethylsulfoxide (C2H6SO) rounded to three significant figures?
1395views - Open Question
Calculate the percentage by mass of lead in Pb(NO3)2.
1771views - Open Question
Calculate the mass percent composition of lithium in Li3PO4.
1459views