3. Chemical Reactions
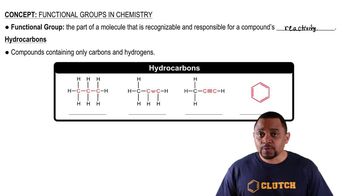
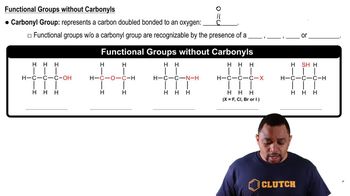
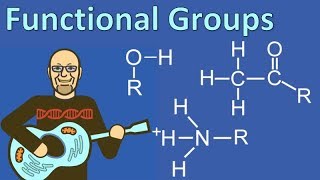
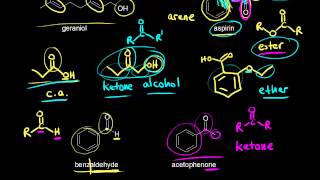
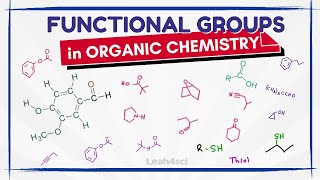
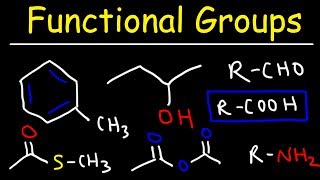
Functional Groups in Chemistry
3. Chemical Reactions
Functional Groups in Chemistry
Showing 6 of 6 videos
Additional 12 creators.
Learn with other creators
Showing 15 of 15 videos
Practice this topic
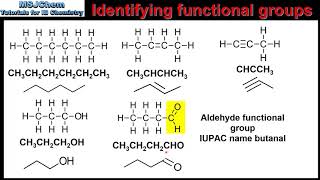
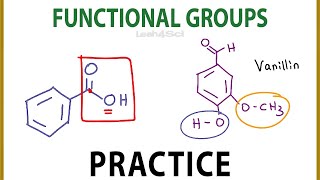
- Multiple ChoiceThe following molecule contains which functional group?
CH3– CH2– CH2– CH2– CH–COOH
1747views - Multiple Choice
Identify the type of functional group(s) present within the following compound.
1671views4rank2comments - Multiple Choice
Identify the type of functional group(s) present within the following compound.
1891views7rank1comments - Multiple ChoiceWhich of the following is an organic compound?1610views
- Open Question
What is the name of the functional group that is attached to this hydrocarbon?
1486views - Open Question
What is the name of the functional group shown in the figure below?
1223views - Open Question
Which molecule shown above has a carbonyl functional group in the form of an aldehyde?
1166views - Open Question
A specific combination of bonded atoms which always react in the same way is known as
1659views