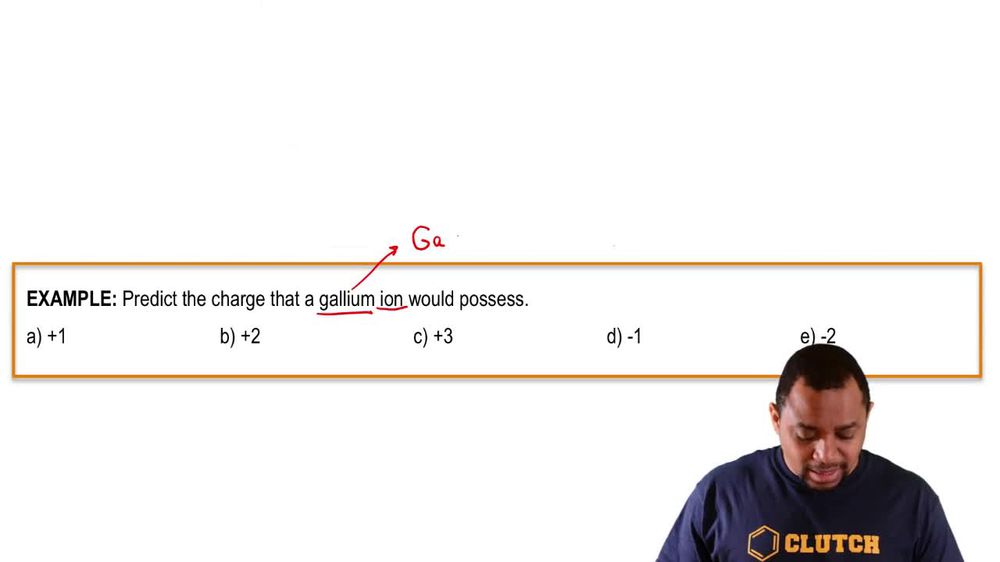
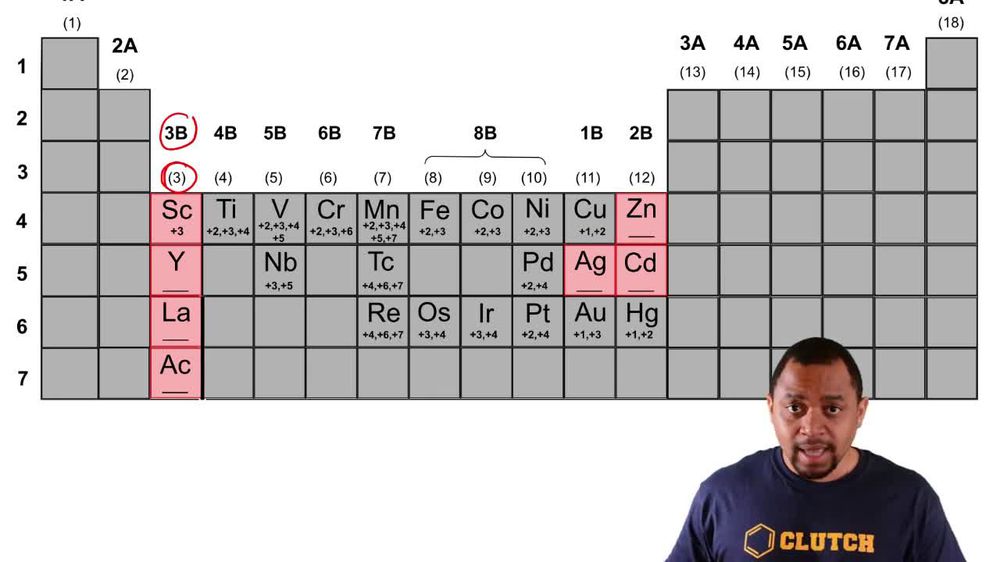
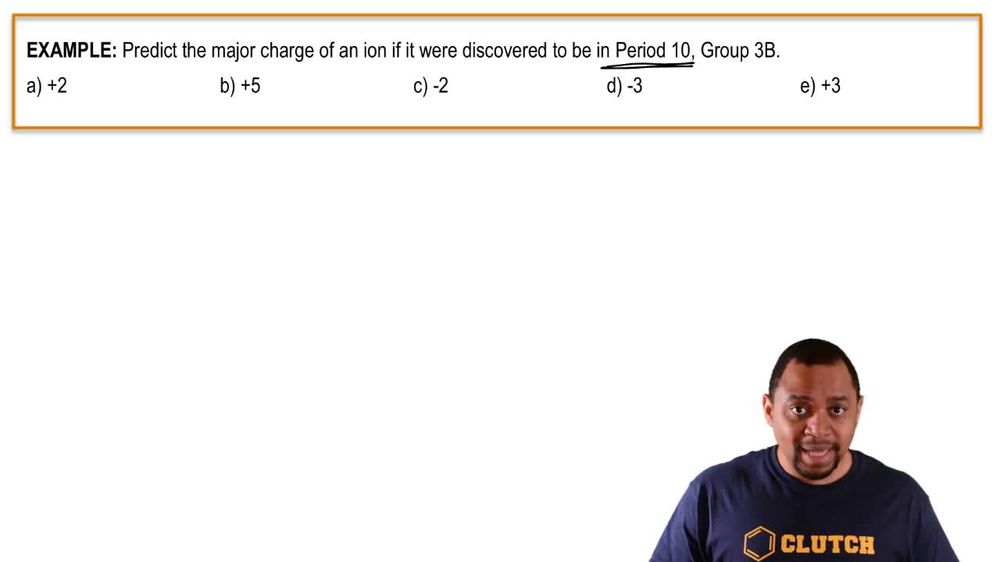2. Atoms & Elements
Periodic Table: Charges
Practice this topic
- Multiple Choice
Which element possesses a -2 charge when it combines with other elements?
1477views14rank - Multiple Choice
What is the likely charge of the element with an atomic number of 47?
2469views12rank - Multiple Choice
How many electrons would the cadmium ion possess?
3576views18rank - Textbook Question
Does the following drawing represent a neutral atom or an ion?
1278views - Textbook Question
Five of the boxes in the following periodic table are colored. Predict the charge on the ion associated with each of these elements.
355views - Textbook Question
Determine the number of valence electrons in each element. Which elements do you expect to lose electrons in chemical reactions? Which do you expect to gain electrons? a. Al b. Sn c. Br d. Se
965views - Textbook Question
Each of the following elements is capable of forming an ion in chemical reactions. By referring to the periodic table, predict the charge of the most stable ion of each: (a) Be (b) Rb (c) As (d) In (e) At.
682views - Multiple ChoiceDo elements in Group 2 tend to lose electrons to form cations with a charge of +2? Because?494views