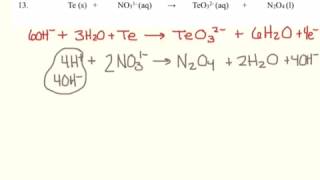
6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Basic Solutions
Practice this topic
- Multiple Choice
Balance the following redox reaction in a basic solution
H2O2 (aq) + ClO2 (aq) → ClO2- (aq) + O2 (g)
2531views5rank1comments - Multiple Choice
Balance the following redox reaction in a basic solution.
ClO2− (aq) → Cl- (aq) + ClO4− (aq)
3539views10rank1comments - Textbook Question
Balance the redox reaction in basic solution. What is the coefficient on the hydroxide ion, and on which side of the equation does it appear? (a) 2 OH- in reactants (b) 4 OH- in products (c) 4 OH- in reactants (d) 3 OH- in reactants
678views - Textbook Question
Complete and balance the following half-reactions. In each case, indicate whether the half-reaction is an oxidation or a reduction. (f) SO32-1aq2 ¡ SO42-1aq2 (basic solution)
620views - Textbook Question
Complete and balance the following half-reactions. In each case indicate whether the half-reaction is an oxidation or a reduction. (g) Cr(OH)3(s) → CrO42-(aq) (basic solution)
1151views - Textbook Question
Complete and balance the following equations, and identify the oxidizing and reducing agents: MnO4-(aq) + Br-(aq) → MnO2(s) + BrO3-(aq) (basic solution)
703views - Multiple ChoiceWhich of the following is the correctly balanced redox reaction for H2O2(aq) + ClO2(aq) → ClO₂⁻(aq) + O₂(g) in a basic solution?445views
- Multiple ChoiceWhich of the following is the correctly balanced redox reaction for MnO4⁻(aq) + Br⁻(aq) → MnO2(s) + BrO3⁻(aq) in a basic solution?519views