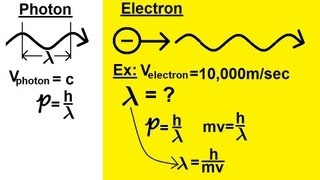
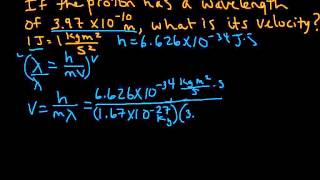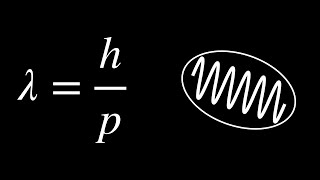9. Quantum Mechanics
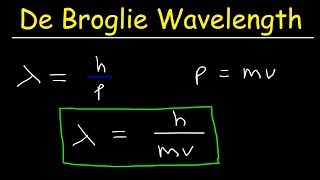
De Broglie Wavelength
Practice this topic
- Multiple Choice
What is the velocity (in m/s) of an electron that has a wavelength of 3.13 x 105 pm? (Mass of an electron = 9.11 x 10-31 kg).
1201views3rank3comments - Multiple Choice
The faster an electron is moving, the _________ its kinetic energy, and the __________ its wavelength.
1352views2rank - Multiple Choice
Consider an atom traveling at 3.00 x 1015 m/s. The de Broglie wavelength is found to be 7.1316 x 10-39. Determine the mass (in g) of the atom.
3144views1rank5comments - Textbook Question
The speed of sound in dry air at 20 °C is 343 m/s and the lowest frequency sound wave that the human ear can detect is approximately 20 Hz. (b) What would be the frequency of electromagnetic radiation with the same wavelength?
5268views - Textbook Question
Use the de Broglie relationship to determine the wavelengths of the following objects: (a) an 85-kg person skiing at 50 km/hr (b) a 10.0-g bullet fired at 250 m/s
695views - Textbook Question
Use the de Broglie relationship to determine the wavelengths of the following objects: (c) a lithium atom moving at 2.5 × 105 m/s (d) an ozone (O3) molecule in the upper atmosphere moving at 550 m/s.
542views - Textbook Question
Among the elementary subatomic particles of physics is the muon, which decays within a few microseconds after formation. The muon has a rest mass 206.8 times that of an electron. Calculate the de Broglie wavelength associated with a muon traveling at 8.85 * 105 cm/s.
640views - Multiple ChoiceA proton in a linear accelerator has a de Broglie wavelength of 132 pm. What is the speed of the proton?331views