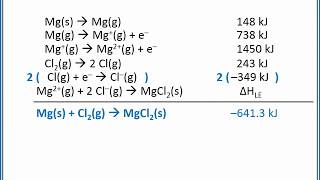11. Bonding & Molecular Structure
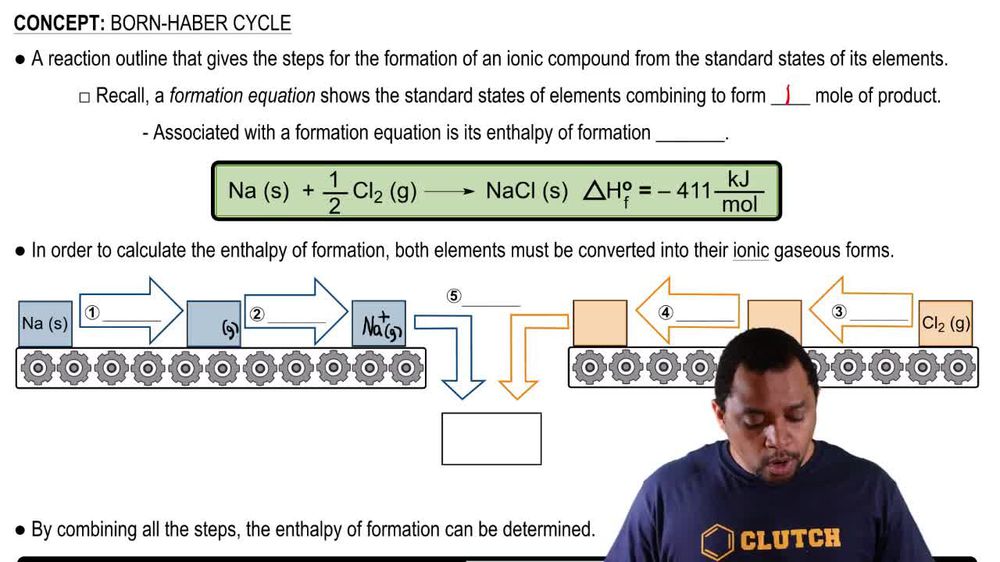
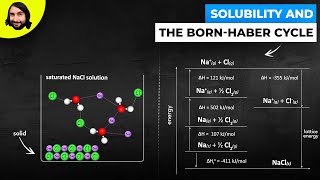
Born Haber Cycle
Practice this topic
- Multiple Choice
Using the Born-Haber Cycle, demonstrate the formation of cesium chloride, CsCl, and calculate its enthalpy of formation.
2424views4rank7comments - Multiple Choice
Calculate the lattice energy for the following formation equation:
2177views3rank4comments - Textbook Question
Given the following information, construct a Born–Haber cycle to calculate the lattice energy of CaCl2(s). (LO 6.13)
Net energy change for the formation of CaCl2(s) form Ca(s) and Cl2(g) = -795.4 kJ/mol
Heat of sublimation for Ca(s) = +178 kJ/mol
Ei1 for Ca(s) = +590 kJ/mol
Ei2 for Ca(g) = +1145 kJ/mol
Bond dissociation energy for Cl2(g) = +243 kJ/mol
Eea1 for Cl(g) = -348.6 kJ/mol
(a) 2603 kJ/mol (b) 2254 kJ/mol (c) 2481 kJ/mo (d) 1663 kJ/mol
707views - Textbook Question
List the individual steps used in constructing a Born–Haber cycle for the formation of BaI2 from the elements. Which of the steps would you expect to be exothermic?
1089views - Textbook Question
Use data from Appendix C, Figure 7.10, and Figure 7.12 to calculate the lattice energy of RbCl.
1040views - Textbook Question
(b) Using data from Appendix C, Figure 7.11, Figure 7.13, and the value of the second ionization energy for Ca, 1145 kJ/mol, calculate the lattice energy of CaCl2.
699views - Multiple ChoiceCalculate the lattice energy for MgO(s) using a Born-Haber cycle and the following information:Mg(s) → Mg(g) +147.1 kJ/molMg(g) → Mg+(g) + e- +737.8 kJ/molMg+(g) → Mg2+(g) + e- +1451 kJ/mol1/2 O2(g) → O(g) +249.0 kJ/molO(g) + e- → O-(g) -141 kJ/molO-(g) + e- → O2-(g) +844 kJ/molMgO(s) → Mg2+(g) + O2-(g) ?754views
- Multiple ChoiceWhich of the following steps is NOT part of the Born-Haber cycle for the formation of lithium bromide (LiBr)?521views