11. Bonding & Molecular Structure
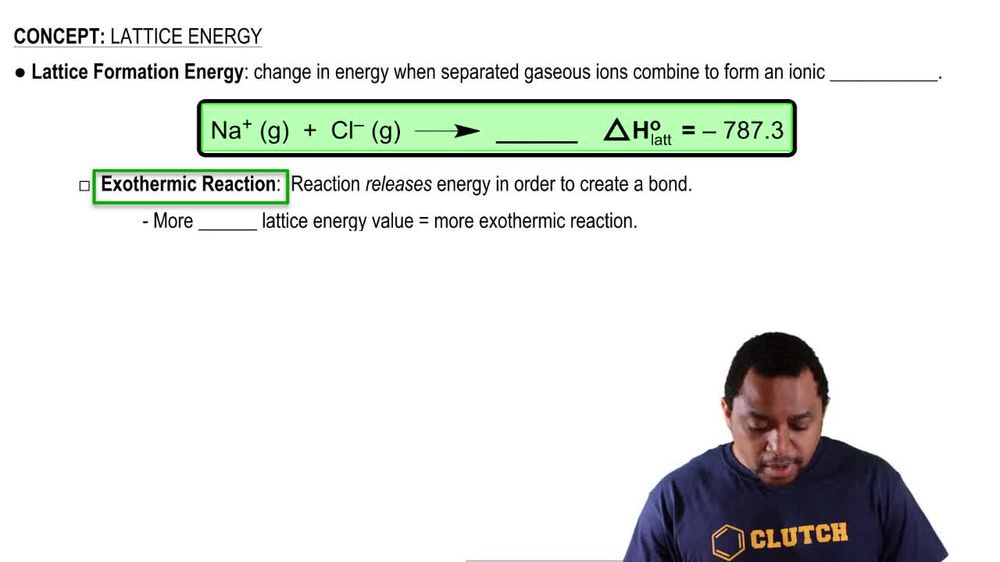
Lattice Energy
Practice this topic
- Multiple Choice
The lattice energy for ionic crystals decreases as the charge of the ions _____ and the size of the ions _____.
1720views1rank - Multiple Choice
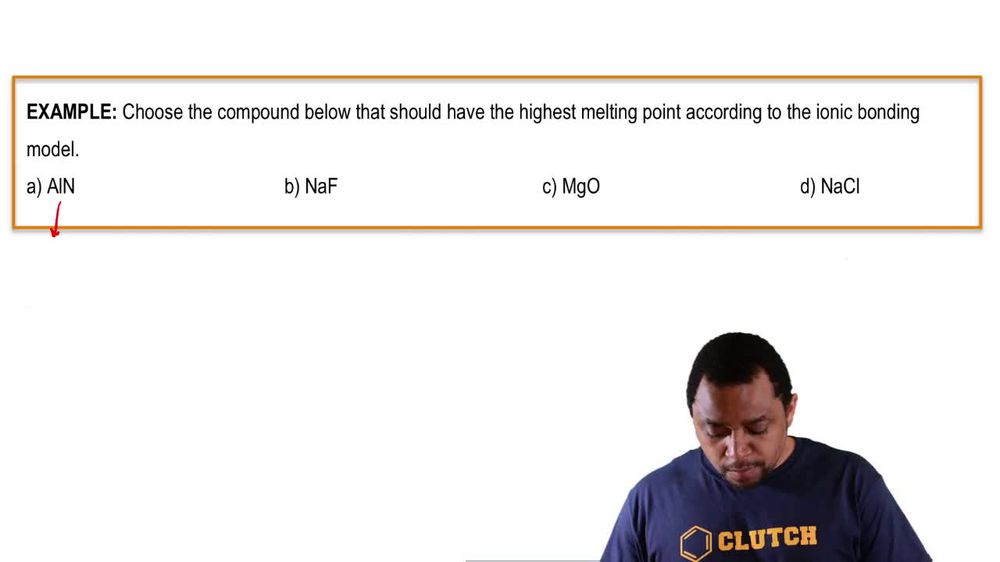
Which of the following compounds would you expect to have the highest boiling point?
1675views6rank - Multiple Choice
The solubilities of CaCrO4 and PbCrO4 in water at 25°C are approximately 0.111 g/L and 0.0905 g/L in H2O respectively. Based on this information, which compound do you think has the smaller lattice energy?
1567views4rank - Multiple ChoiceWhich of the following reactions has ΔHrxn = ΔHlattice?1169views
- Textbook Question
Illustrated are four ions — A, B, X, and Y— showing their relative ionic radii. The ions shown in red carry positive charges: a 2+ charge for A and a 1+ charge for B. Ions shown in blue carry negative charges: a 1- charge for X and a 2- charge for Y. (b) Among the combinations in part (a), which leads to the ionic compound having the largest lattice energy?
594views - Textbook Question
A portion of a two-dimensional 'slab' of NaCl(s) is shown here (see Figure 8.2) in which the ions are numbered. (d) Consider ion 5. How many repulsive interactions are shown for it?
513views - Textbook Question
(c) Would you expect salts like NaCl, which have singly charged ions, to have larger or smaller lattice energies compared to salts like CaO which are composed of doubly-charged ions?
471views - Textbook Question
(b) Write the chemical equation that represents the process of lattice energy for the case of NaCl.
1771views