11. Bonding & Molecular Structure
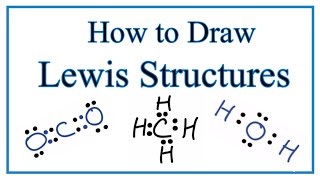
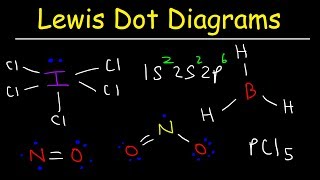
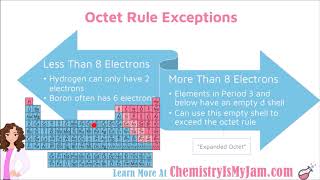
Lewis Dot Structures: Exceptions
Practice this topic
- Multiple Choice
Determine the Lewis Dot Structure for the following compound:SOCl2
3079views4rank3comments - Multiple Choice
Draw the Lewis Dot Structure for the radical hydroxide, OH.
4123views6rank1comments - Multiple Choice
Draw the Lewis Dot Structure for POCl3.
5464views8rank1comments - Multiple Choice
Metalloids can sometimes adopt the bonding preferences of similar nonmetals. Based on your knowledge of expanded octets, draw the Lewis Dot Structure for the following ion, SiF62–.
3611views6rank1comments - Textbook Question
Using Lewis symbols and Lewis structures, diagram the formation of PF3 from P and F atoms, showing valence-shell electrons. (e) Does PF3 obey the octet rule?
552views - Textbook Question
Draw Lewis structures for the following: (a) CH2Cl2 (b) ClCN (c) SF2
617views - Textbook Question
(b) Which of these compounds or ions is an exception to the octet rule: borohydride (BH4-), borazine (B3N3H6, which is analogous to benzene with alternating B and N in the ring), or boron trichloride?
974views - Textbook Question
Draw the Lewis structures for each of the following molecules or ions. Identify instances where the octet rule is not obeyed; state which atom in each compound does not follow the octet rule; and state how many electrons surround these atoms: (a) PF6-, (b) BeCl2, (c) NH3, (d) XeF2O (the Xe is the central atom), (e) SO42- .
1336views