Table of contents
- 1. Introduction to Biology
- 2. Chemistry
- 3. Water
- 4. Biomolecules
- 5. Cell Components
- 6. The Membrane
- 7. Energy and Metabolism
- 8. Respiration
- 9. Photosynthesis
- 10. Cell Signaling
- 11. Cell Division
- 12. Meiosis
- 13. Mendelian Genetics
- Introduction to Mendel's Experiments
- Genotype vs. Phenotype
- Punnett Squares
- Mendel's Experiments
- Mendel's Laws
- Monohybrid Crosses
- Test Crosses
- Dihybrid Crosses
- Punnett Square Probability
- Incomplete Dominance vs. Codominance
- Epistasis
- Non-Mendelian Genetics
- Pedigrees
- Autosomal Inheritance
- Sex-Linked Inheritance
- X-Inactivation
- 14. DNA Synthesis
- 15. Gene Expression
- 16. Regulation of Expression
- Introduction to Regulation of Gene Expression
- Prokaryotic Gene Regulation via Operons
- The Lac Operon
- Glucose's Impact on Lac Operon
- The Trp Operon
- Review of the Lac Operon & Trp Operon
- Introduction to Eukaryotic Gene Regulation
- Eukaryotic Chromatin Modifications
- Eukaryotic Transcriptional Control
- Eukaryotic Post-Transcriptional Regulation
- Eukaryotic Post-Translational Regulation
- 17. Viruses
- 18. Biotechnology
- 19. Genomics
- 20. Development
- 21. Evolution
- 22. Evolution of Populations
- 23. Speciation
- 24. History of Life on Earth
- 25. Phylogeny
- 26. Prokaryotes
- 27. Protists
- 28. Plants
- 29. Fungi
- 30. Overview of Animals
- 31. Invertebrates
- 32. Vertebrates
- 33. Plant Anatomy
- 34. Vascular Plant Transport
- 35. Soil
- 36. Plant Reproduction
- 37. Plant Sensation and Response
- 38. Animal Form and Function
- 39. Digestive System
- 40. Circulatory System
- 41. Immune System
- 42. Osmoregulation and Excretion
- 43. Endocrine System
- 44. Animal Reproduction
- 45. Nervous System
- 46. Sensory Systems
- 47. Muscle Systems
- 48. Ecology
- 49. Animal Behavior
- 50. Population Ecology
- 51. Community Ecology
- 52. Ecosystems
- 53. Conservation Biology
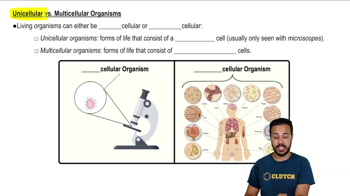
1. Introduction to Biology
Introduction to Biology
1. Introduction to Biology
Introduction to Biology
+2
Additional 2 creators.
Learn with other creators
Showing 5 of 5 videos
Practice this topic
- Multiple Choice
The area of science that studies life and its processes is called:
20360views496rank - Multiple Choice
All living organisms are ___________:
17072views171rank4comments - Multiple ChoiceHow do heterotrophs obtain energy?259views
- Multiple ChoiceWhich of the following is NOT one of the six primary taste sensations?231views