7. Energy and Metabolism
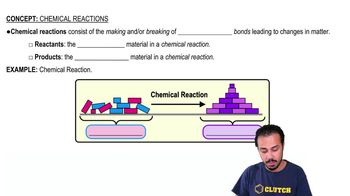
Chemical Reactions
7. Energy and Metabolism
Chemical Reactions
Additional 3 creators.
Learn with other creators
Showing 6 of 6 videos
Practice this topic
- Multiple Choice
Which of the following statements is true for all exergonic reactions?
8405views95rank - Multiple ChoiceWhich statement is true about chemical reactions?4819views
- Multiple ChoiceWhich statement describes a reversible reaction that has reached chemical equilibrium?3153views1rank
- Multiple ChoiceAn exergonic (spontaneous) reaction is a chemical reaction that __________.3003views