14. Solutions
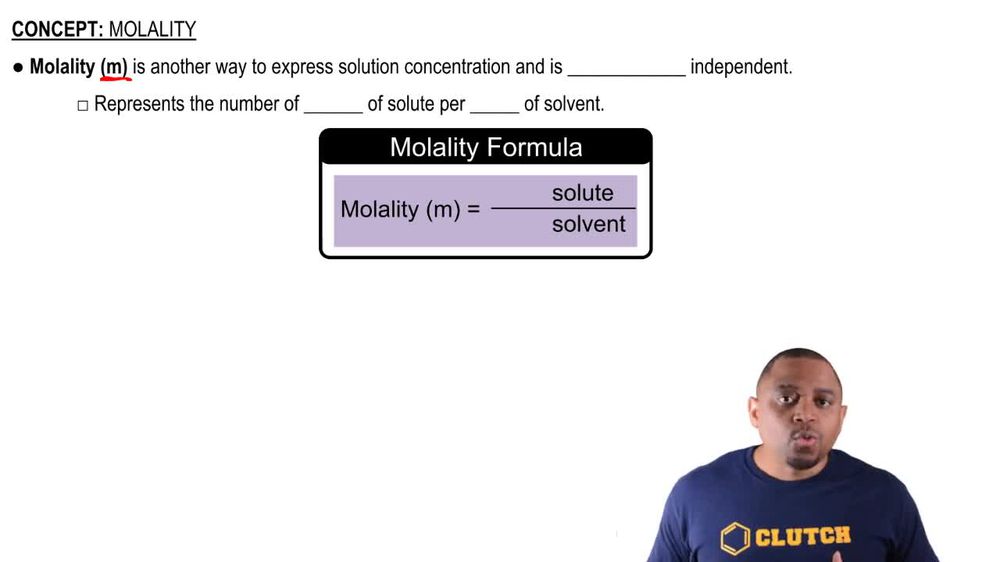
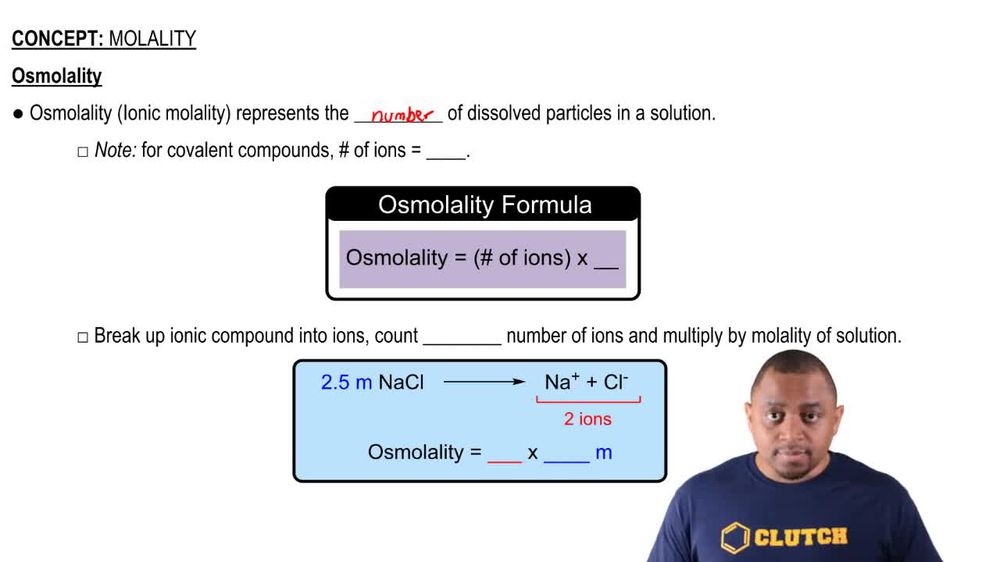
Molality
Practice this topic
- Multiple Choice
A solution is prepared by dissolving 43.0 g potassium chlorate, KClO3, in enough water to make 100.0 mL of solution. If the density of the solution is 1.760 g/mL, what is the molality of KClO3 in the solution?
2279views14rank - Multiple Choice
The density of a 15.7 M methanol (CH3OH) solution is 0.858 g/mL. If H2O is the solvent, what is the molality of the solution?
2740views13rank4comments - Multiple Choice
What is the ionic molality of sodium ions in a solution of 25.7 g NaNO3 dissolved in enough water to make a 150.0 mL of solution? Density of the solution is 1.02 g/mL.
1793views12rank4comments - Multiple ChoiceWhat is the molarity of a solution prepared by dissolving 5.00 g of NaCl in 500.0 mL of water?1242views
- Textbook Question
The figure shows two identical volumetric flasks containing the same solution at two temperatures. (b) Does the molality of the solution change with the change in temperature? [Section 13.4]
504views - Textbook Question
A solution is made containing 20.8 g of phenol (C6H5OH) in 425 g of ethanol (CH3CH2OH). Calculate (c) the molality of phenol.
1021views - Textbook Question
Ascorbic acid (vitamin C, C6H8O6) is a water-soluble vitamin. A solution containing 80.5 g of ascorbic acid dissolved in 210 g of water has a density of 1.22 g/mL at 55 °C. Calculate (c) the molality,
565views - Textbook Question
The density of acetonitrile (CH3CN) is 0.786 g/mL and the density of methanol (CH3OH) is 0.791 g/mL. A solution is made by dissolving 22.5 mL of CH3OH in 98.7 mL of CH3CN. (b) What is the molality of the solution?
1014views