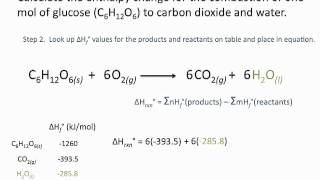8. Thermochemistry
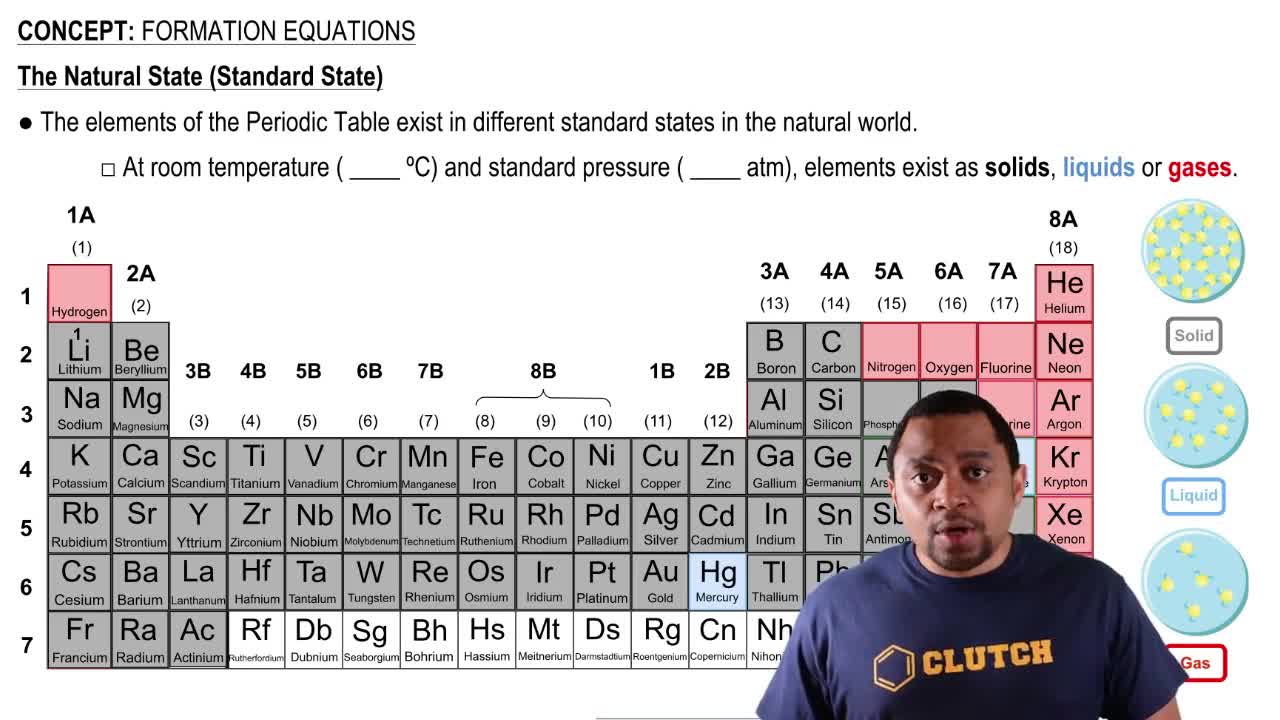
Formation Equations
Practice this topic
- Multiple Choice
Which of the following represents the formation equation for barium nitrate, Ba(NO3)2?
1338views7rank - Multiple Choice
Identify a substance that is not in its standard state.
2143views - Multiple Choice
Ibuprofen is used as an anti-inflammatory agent used to deal with pain and bring down fevers. If it has a molecular formula of C13H18O2, determine the balanced chemical equation that would give you directly the enthalpy of formation for ibuprofen.
1500views6rank - Textbook Question
(c) What is meant by the term standard enthalpy of formation?
456views - Textbook Question
(a) Why does the standard enthalpy of formation of both the very reactive fluorine (F2) and the almost inert gas nitrogen (N2) both read zero?
556views - Textbook Question
For each of the following compounds, write a balanced thermochemical equation depicting the formation of one mole of the compound from its elements in their standard states and then look up ΔH°f for each substance in Appendix C. (a) NO2(g) (b) SO3(g) (c) NaBr(s) (d) Pb(NO3)2(s).
1050views - Textbook Question
Write balanced equations that describe the formation of the following compounds from elements in their standard states, and then look up the standard enthalpy of formation for each substance in Appendix C: (a) CH3OH(l)
1009views1rank - Multiple ChoiceWhich of the following is the correct chemical equation for the formation of BF3(g) from its elements in their standard states?688views