11. Bonding & Molecular Structure
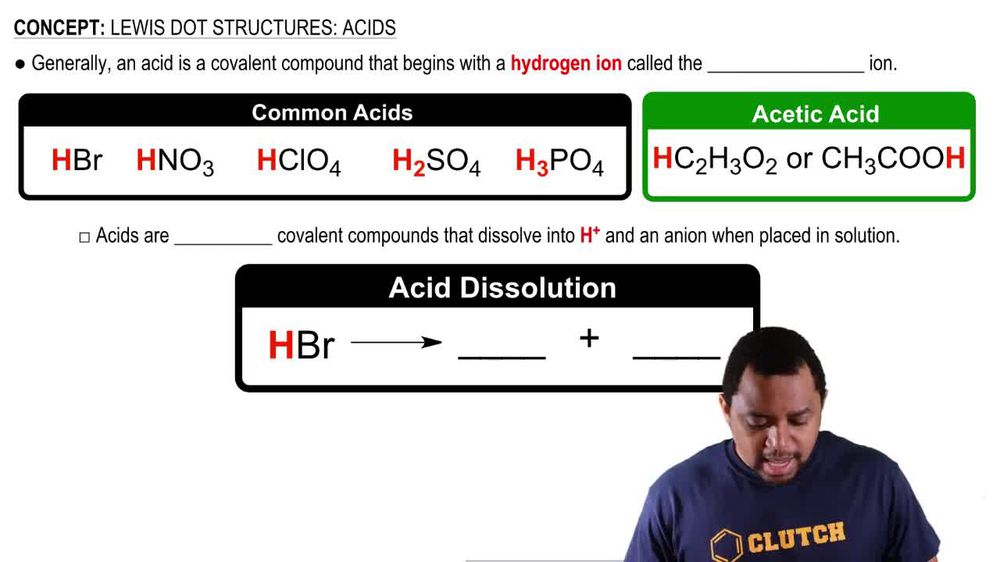
Lewis Dot Structures: Acids
Practice this topic
- Multiple Choice
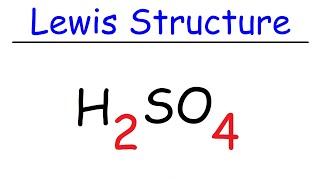
Determine the Lewis Dot Structure for the following acid:H2SO4
3805views4rank - Multiple Choice
Determine the Lewis Dot Structure for hydrocyanic acid:HCN
1960views5rank1comments - Multiple Choice
Determine the Lewis Dot Structure for chloric acid:HClO3
5404views2rank3comments - Multiple Choice
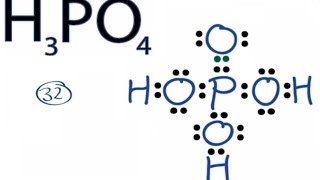
Determine the Lewis Dot Structure for phosophoric acid:H3PO4
4282views5rank1comments - Textbook Question
There are many Lewis structures you could draw for sulfuric acid, H2SO4 (each H is bonded to an O). (a) What Lewis structure(s) would you draw to satisfy the octet rule?
646views - Textbook Question
A common form of elemental phosphorus is the tetrahedral P4 molecule, where all four phosphorus atoms are equivalent:
Draw a Lewis structure for a linear P4 molecule that satisfies the octet rule. Does this molecule have resonance structures?
1243views - Textbook Question
A common form of elemental phosphorus is the tetrahedral P4 molecule, where all four phosphorus atoms are equivalent:
(b) How many P-P bonds are there in the molecule?
765views - Textbook Question
Draw the Lewis structure for each compound. c. H3AsO4
1204views