Acetylene (C2H2) and nitrogen (N2) both contain a triple bond, but they differ greatly in their chemical properties. (e) Both N2 and C2H2 possess triple bonds with quite high bond enthalpies (Table 8.3). Calculate the enthalpy of hydrogenation per mole for both compounds: acetylene plus H2 to make methane, CH4; nitrogen plus H2 to make ammonia, NH3.

A common form of elemental phosphorus is the tetrahedral P4 molecule, where all four phosphorus atoms are equivalent:
(b) How many P-P bonds are there in the molecule?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
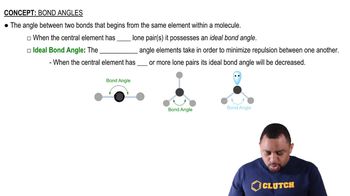
Key Concepts
Molecular Structure

Covalent Bonds
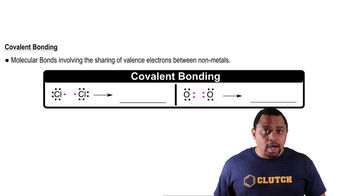
Counting Bonds
Under special conditions, sulfur reacts with anhydrous liquid ammonia to form a binary compound of sulfur and nitrogen. The compound is found to consist of 69.6% S and 30.4% N. Measurements of its molecular mass yield a value of 184.3 g/mol. The compound occasionally detonates on being struck or when heated rapidly. The sulfur and nitrogen atoms of the molecule are joined in a ring. All the bonds in the ring are of the same length. (a) Calculate the empirical and molecular formulas for the substance.
Under special conditions, sulfur reacts with anhydrous liquid ammonia to form a binary compound of sulfur and nitrogen. The compound is found to consist of 69.6% S and 30.4% N. Measurements of its molecular mass yield a value of 184.3 g/mol. The compound occasionally detonates on being struck or when heated rapidly. The sulfur and nitrogen atoms of the molecule are joined in a ring. All the bonds in the ring are of the same length. (b) Write Lewis structures for the molecule, based on the information you are given. (Hint: You should find a relatively small number of dominant Lewis structures.)
A common form of elemental phosphorus is the tetrahedral P4 molecule, where all four phosphorus atoms are equivalent:
Draw a Lewis structure for a linear P4 molecule that satisfies the octet rule. Does this molecule have resonance structures?
Trifluoroacetic acid has the chemical formula CF3CO2H. It is a colorless liquid that has a density of 1.489 g/mL (a) Trifluoroacetic acid contains one CF3 unit and is connected to the other C atom which bonds with both O’s. Draw the Lewis structure for trifluoroacetic acid.
Trifluoroacetic acid has the chemical formula CF3CO2H. It is a colorless liquid that has a density of 1.489 g/mL. (b) Trifluoroacetic acid can react with NaOH in aqueous solution to produce the trifluoroacetate ion, CF3COO2. Write the balanced chemical equation for this reaction.
