11. Bonding & Molecular Structure
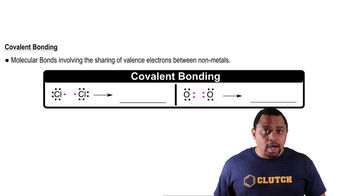
Chemical Bonds
11. Bonding & Molecular Structure
Chemical Bonds
Showing 7 of 7 videos
Additional 4 creators.
Learn with other creators
Showing 7 of 7 videos
Practice this topic
- Multiple Choice
The strength of an ionic bond comes principally from:
2818views26rank - Multiple Choice
Which of the following is not a physical property attributed to metallic bonding?
2174views15rank - Multiple Choice
Which of the following statements is true?
3098views23rank1comments - Multiple ChoiceWhich best describes metallic bonding?2021views
- Open Question
Which of the following can easily form an ionic bond with a cation? Sr, Ne, NH4+, PO43−
1453views - Open Question
A(n) _____ refers to two or more atoms held together by covalent bonds.
1843views - Open Question
A molecule of water (H2O) is formed by what type of bond?
1552views - Open Question
A phrase that applies to covalent bonding and not other kinds of bonds is...
1778views