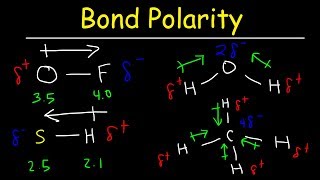
11. Bonding & Molecular Structure
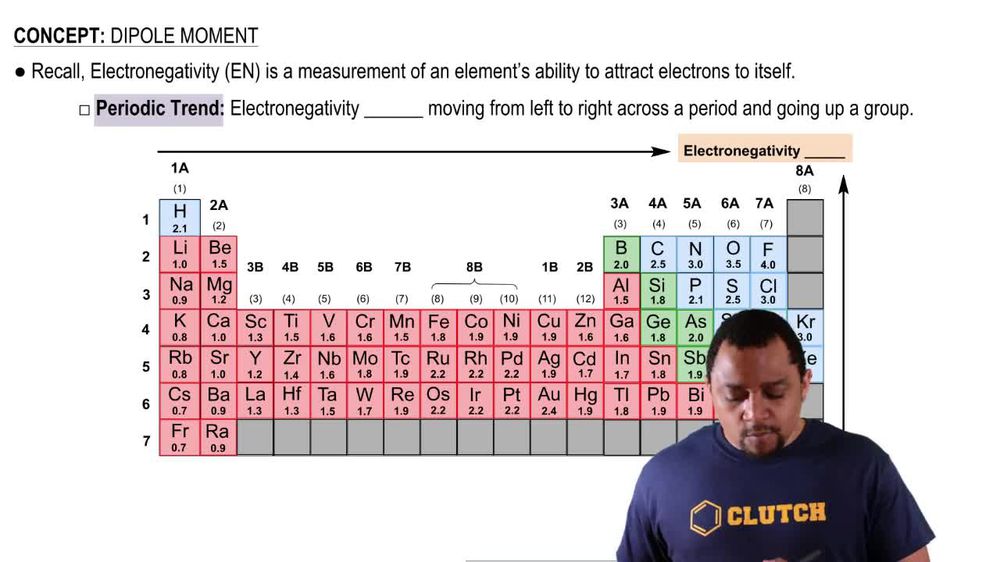
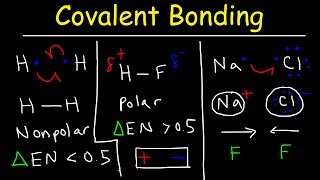
Dipole Moment
Practice this topic
- Multiple Choice
Which of the following correctly identifies the chemical bond between a carbon and oxygen atom?
3582views17rank - Multiple ChoiceWhich of the following is the most polar bond?1415views
- Multiple ChoiceWhat is the percent ionic character of a 125 pm bond which has a bond dipole of 4.5 D?1360views
- Multiple Choice
Arrange the following molecules in order of decreasing dipole moment.
H–I, H–F, H–Br, H–Cl
1250views11rank2comments - Textbook Question
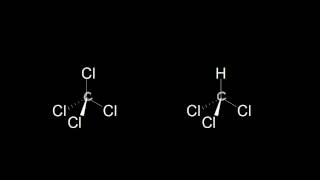
Two dichloroethylene molecules with the same chemical formula 1C2H2Cl22, but different arrangements of atoms are shown. (b) Which form of dichloroethylene has a dipole moment of 2.39 D, and which has a dipole moment of 0.00 D?
372views - Textbook Question
(a) Does CS2 have a dipole moment? If so, in which direction does the net dipole point? (b) Does SO2 have a dipole moment? If so, in which direction does the net dipole point?
861views - Textbook Question
(b) It turns out that ozone, O3, has a small dipole moment. How is this possible, given that all the atoms are the same?
1662views - Textbook Question
(c) Does the molecule BF2Cl have a dipole moment?
1411views