Textbook Question
(a) Does CS2 have a dipole moment? If so, in which direction does the net dipole point? (b) Does SO2 have a dipole moment? If so, in which direction does the net dipole point?
861
views

 Verified step by step guidance
Verified step by step guidance
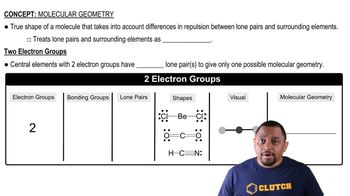


(a) Does CS2 have a dipole moment? If so, in which direction does the net dipole point? (b) Does SO2 have a dipole moment? If so, in which direction does the net dipole point?
(b) If you react BF3 to make the ion BF32-, is this ion planar?
(c) Does the molecule BF2Cl have a dipole moment?
(a) Consider the following two molecules: PCl3 and BCl3. Which molecule has a nonzero dipole moment?