14. Solutions
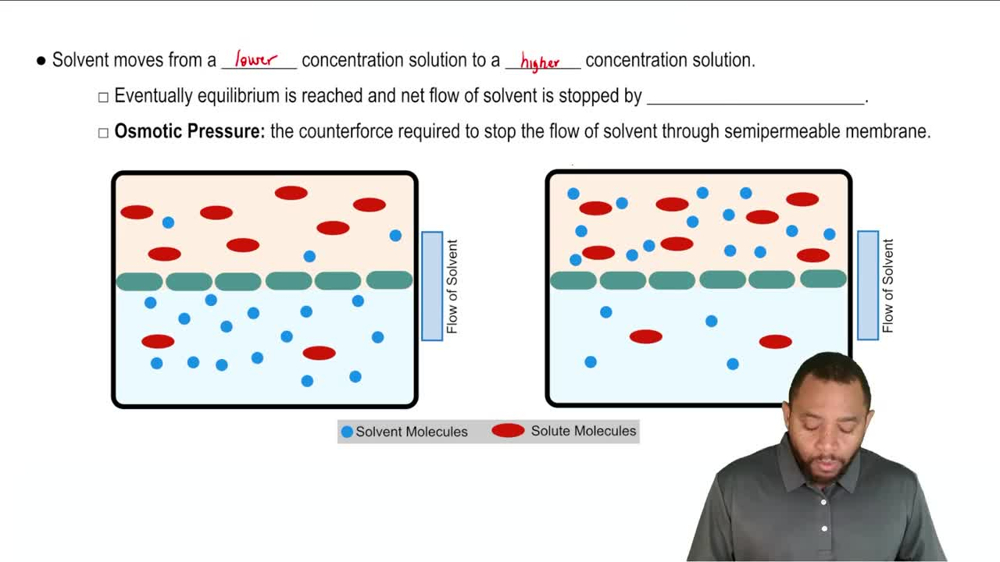
Osmosis
Practice this topic
- Multiple Choice
A semipermeable membrane is placed between the following solutions.
Which solution will increase in volume?
1917views3rank - Multiple Choice
Four U tubes each have distilled water in the right arm, a solution in the left arm, and a semipermeable membrane between the arms. If the solute is LiF, which solution is most concentrated?
1567views3comments - Multiple Choice
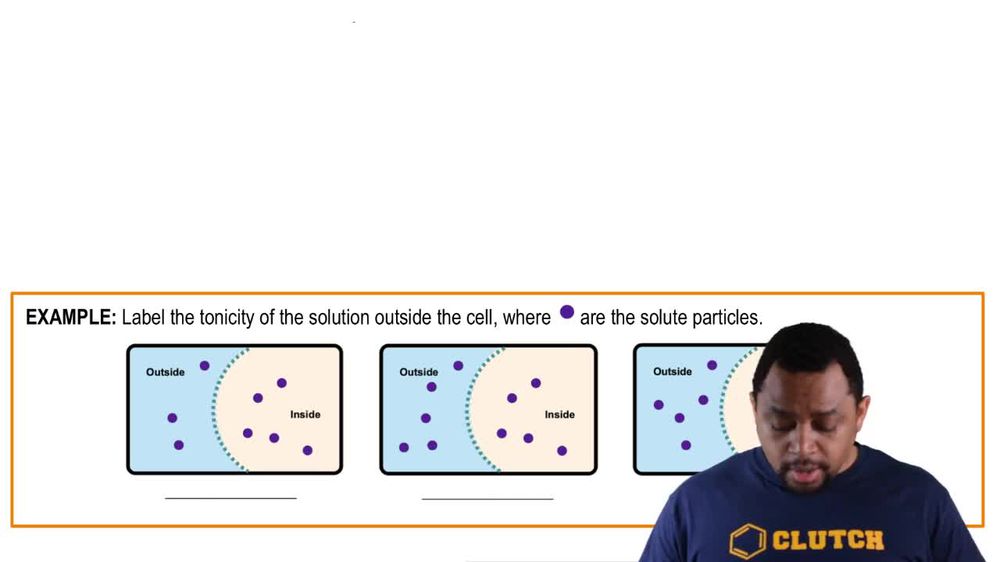
Identify the direction of water flow between 2 solutions separates by semipermeable membrane, where are the solute particles.
1457views2comments - Multiple Choice
If the fluid surrounding a patient's red blood cells is depleted in electrolytes, is crenation or hemolysis more likely to occur?
1607views2comments - Textbook Question
Suppose you had a balloon made of some highly flexible semipermeable membrane. The balloon is filled completely with a 0.2 M solution of some solute and is submerged in a 0.1 M solution of the same solute:
Initially, the volume of solution in the balloon is 0.25 L. Assuming the volume outside the semipermeable membrane is large, as the illustration shows, what would you expect for the solution volume inside the balloon once the system has come to equilibrium through osmosis? [Section 13.5]
451views - Textbook Question
The first stage of treatment at the reverse osmosis plant in Carlsbad, California, is to flow the water through rock, sand, and gravel as shown here. Would this step remove particulate matter? Would this step remove dissolved salts?
[Section 18.4]
110views - Open Question
What would happen if the tubing with the yellow band was placed in a beaker of distilled water?
883views - Textbook Question
The first stage of treatment at a reverse osmosis plant is to flow the water through rock, sand, and gravel as shown here. Would this step remove particulate matter? Would this step remove dissolved salts?
1views