Draw the structures of the following nucleotides.
(c) cyclic guanosine monophosphate (cGMP)

 Verified step by step guidance
Verified step by step guidance


Draw the structures of the following nucleotides.
(c) cyclic guanosine monophosphate (cGMP)
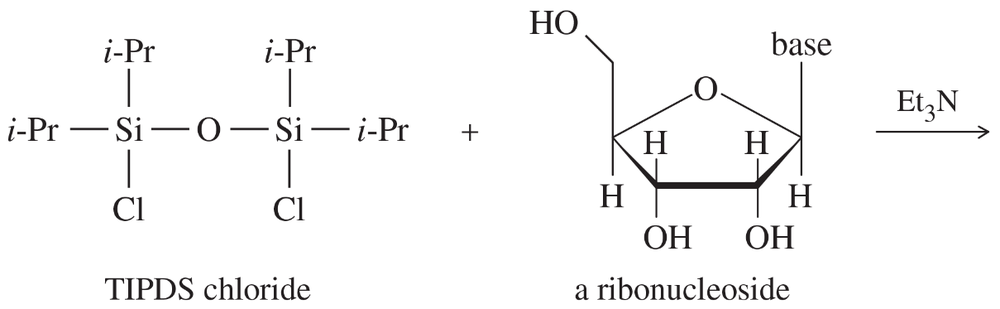
Some protecting groups can block two OH groups of a carbohydrate at the same time. One such group is shown here, protecting the 4-OH and 6-OH groups of β-d-glucose.
(a) What type of functional group is involved in this blocking group?
(b) What did glucose react with to form this protected compound?
(c) When this blocking group is added to glucose, a new chiral center is formed. Where is it? Draw the stereoisomer that has the other configuration at this chiral center. What is the relationship between these two stereoisomers of the protected compound?
(d) Which of the two stereoisomers in part (c) do you expect to be the major product? Why?
Draw the structures of the following nucleotides.
(a) guanosine triphosphate (GTP)
When the gum of the shrub Sterculia setigera is subjected to acidic hydrolysis, one of the water-soluble components of the hydrolysate is found to be tagatose. The following information is known about tagatose:
(1) Molecular formula C6H12O6
(2) Undergoes mutarotation.
(3) Does not react with bromine water.
(4) Reduces Tollens reagent to give D-galactonic acid and D-talonic acid.
(5) Methylation of tagatose (using excess CH3I and Ag2O) followed by acidic hydrolysis gives 1,3,4,5-tetra-O-methyltagatose.
(a) Draw a Fischer projection structure for the open-chain form of tagatose.
Draw the structures of the following nucleotides.
(b) deoxycytidine monophosphate (dCMP)
Show what product results if the aldopentose formed from degradation of X is further degraded to an aldotetrose. Does HNO3 oxidize this aldotetrose to an optically active aldaric acid?