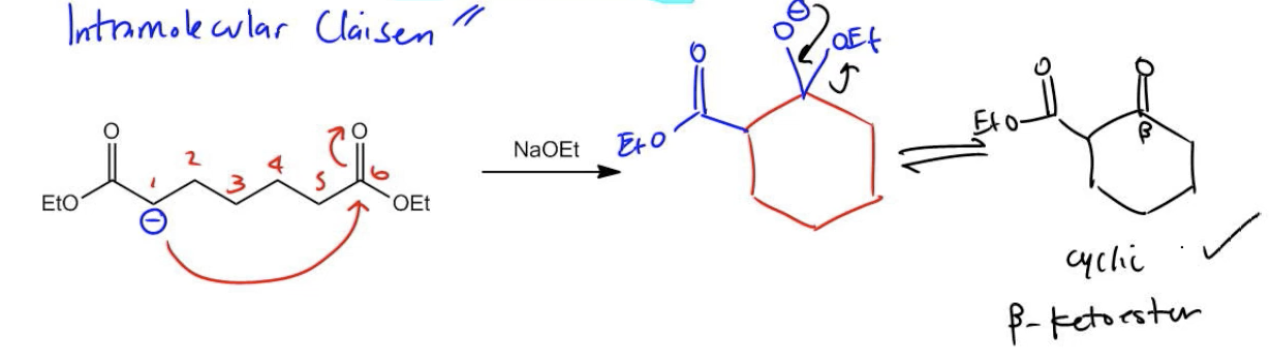
Intramolecular condensation is a specialized reaction that occurs when dicarbonyl compounds, such as diketones, diesters, or dialdehydes, self-condense to form cyclic structures. The formation of 5 or 6-membered rings is particularly favorable due to their stability, while smaller rings (3 or 4 members) are generally too unstable to form without significant energy input.
To initiate this reaction, an enolate must be formed at a position that allows for the creation of a stable ring. For instance, in a diketone, there are typically multiple positions available for enolate formation. However, the choice of position is crucial. If the enolate is formed at carbon 2, the resulting ring would only consist of 4 atoms, which is insufficient for stability. Conversely, forming the enolate at carbon 1 allows for a 6-membered ring to form, which is more favorable.
The mechanism of this reaction follows the typical aldol reaction pathway. The enolate acts as a nucleophile, attacking the carbonyl carbon, leading to the formation of a beta-hydroxycarbonyl compound. This compound can then undergo dehydration, resulting in a cyclic enone, which is a stable product of the intramolecular aldol reaction. The final structure is characterized by a cyclic enone, confirming the reaction's classification as an intramolecular aldol condensation.
In summary, the key to successful intramolecular condensation lies in the formation of stable 5 or 6-membered rings through careful selection of enolate formation positions, leading to the production of stable cyclic enones.