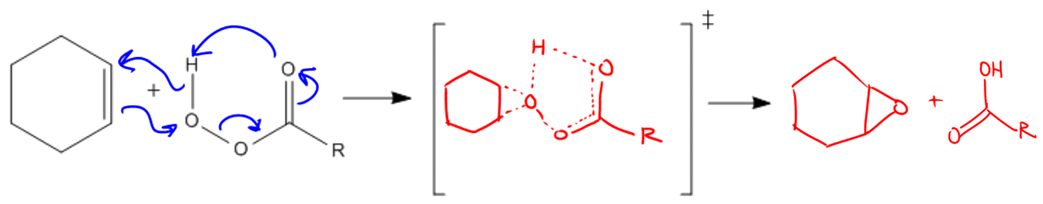
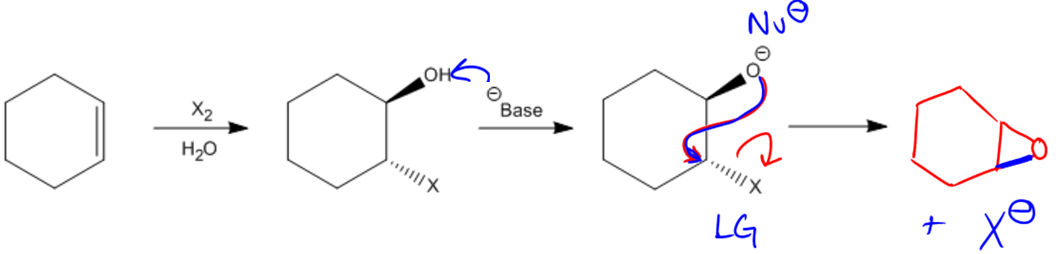
Epoxidation is a specific addition reaction that introduces oxygen to a double bond, resulting in the formation of a new functional group known as an epoxide. An epoxide is characterized as a cyclic three-membered ether, which can be understood as a structure where an oxygen atom is bonded to two carbon groups (R) that are also connected to each other, forming a closed ring. This unique structure distinguishes epoxides from regular ethers, which have a linear arrangement.
The process of epoxidation involves the addition of one oxygen atom to a double bond, transforming it into this three-membered ring structure. To achieve this transformation, a class of compounds known as peroxy acids is utilized. Peroxy acids share a structural similarity with carboxylic acids, differing primarily by the presence of an additional oxygen atom. The general formula for a peroxy acid can be expressed as RCO₃H, where R represents the hydrocarbon chain. In contrast, a carboxylic acid has the formula RCO₂H.
Common examples of peroxy acids used in epoxidation reactions include MCPBA (meta-Chloroperoxybenzoic acid) and MMPP (m-Methyl-m-chloroperoxybenzoic acid). While the specific structures of these peroxy acids are not essential to memorize, recognizing them as peroxy acids is important for understanding their role in the epoxidation process.