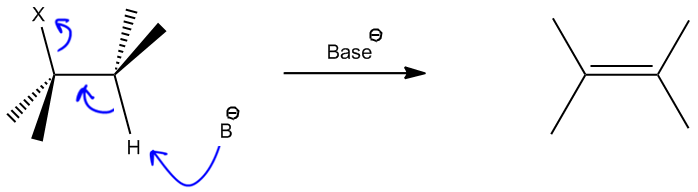
Dehydrohalogenation is a specific type of elimination reaction that follows the E2 mechanism, where a hydrogen atom and a halogen atom are removed from an alkyl halide. This process is characterized by the breaking of two sigma bonds and the formation of a pi bond, resulting in the creation of a double bond.
In an E2 reaction, the structure of the alkyl halide plays a crucial role. Generally, more substituted alkyl halides, such as secondary or tertiary halides, are preferred because they favor elimination. The reaction requires a strong base, which can influence the product formed. Depending on the base used, the reaction may lead to either Zaitsev or Hofmann products, with Zaitsev's rule indicating that the more substituted alkene is typically favored.
During the dehydrohalogenation process, the base abstracts a beta hydrogen, which is crucial for the elimination to occur. The beta hydrogen must be in an anti-coplanar position relative to the leaving group (the halogen), ensuring that the groups are oriented favorably for elimination. This orientation can be visualized using a Newman projection, where the groups are positioned opposite each other.
The mechanism involves three key steps: the base removes the beta hydrogen, the electrons from the C-H bond form a double bond, and the leaving group (X) is expelled. The result is a new alkene, along with the conjugate acid of the base and the halide ion.
Understanding the concepts of anti-coplanar geometry, the roles of different bases, and the implications of Zaitsev and Hofmann rules is essential for mastering dehydrohalogenation. Practicing with various reagents will help solidify your grasp of this important reaction mechanism.