Carbohydrate chemistry, often referred to as sugar chemistry, encompasses the study of sugars, saccharides, and carbohydrates, which are all terms for the same class of molecules. The term "carbohydrate" literally means a hydrate of carbon, indicating that these molecules consist of carbon atoms combined with water. In chemical terms, a carbohydrate can be represented by the general formula \( C_n(H_2O)_n \), where \( n \) is a number greater than or equal to 3, signifying that the smallest sugar, or monosaccharide, contains at least three carbon atoms.
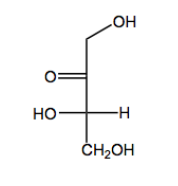
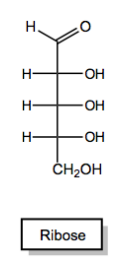
Monosaccharides, the simplest form of carbohydrates, can exist in either straight-chain or cyclic forms. Each monosaccharide must have one oxygen atom attached to every carbon atom, ensuring a one-to-one ratio of oxygen to carbon. Additionally, monosaccharides begin their existence as either aldehydes or ketones. Aldehyde-containing sugars are termed aldoses, while those with ketones are called ketoses. For example, glucose is an aldose, while fructose is a ketose.
When discussing the structure of monosaccharides, it is essential to recognize their stereochemistry. The total number of stereoisomers for a sugar can be calculated using the formula \( 2^n \), where \( n \) represents the number of chiral centers. For instance, glucose has four chiral centers, leading to \( 2^4 = 16 \) possible stereoisomers. This complexity is further illustrated by the concept of epimers, which are specific types of diastereomers that differ at only one chiral carbon. Understanding these relationships is crucial for grasping the intricate nature of carbohydrate structures.
In terms of naming, monosaccharides can be categorized by their carbonyl type (aldehyde or ketone) and the length of their carbon chain. For example, a five-carbon sugar is referred to as a pentose, while a six-carbon sugar is called a hexose. However, some names, such as "tetraose" for a four-carbon sugar, do not follow the expected IUPAC conventions due to historical naming practices.
As you delve deeper into carbohydrate chemistry, it is important to familiarize yourself with the various terms and concepts, including the structural representations of sugars, their functional groups, and the implications of their stereochemistry. This foundational knowledge will serve as a stepping stone for more advanced topics in the field.