- 1. Matter and Measurements4h 29m
- What is Chemistry?7m
- The Scientific Method9m
- Classification of Matter16m
- States of Matter8m
- Physical & Chemical Changes19m
- Chemical Properties8m
- Physical Properties5m
- Intensive vs. Extensive Properties13m
- Temperature (Simplified)9m
- Scientific Notation13m
- SI Units (Simplified)5m
- Metric Prefixes24m
- Significant Figures (Simplified)11m
- Significant Figures: Precision in Measurements7m
- Significant Figures: In Calculations19m
- Conversion Factors (Simplified)15m
- Dimensional Analysis22m
- Density12m
- Specific Gravity9m
- Density of Geometric Objects19m
- Density of Non-Geometric Objects8m
- 2. Atoms and the Periodic Table5h 22m
- The Atom (Simplified)9m
- Subatomic Particles (Simplified)12m
- Isotopes17m
- Ions (Simplified)22m
- Atomic Mass (Simplified)18m
- Atomic Mass (Conceptual)12m
- Periodic Table: Element Symbols6m
- Periodic Table: Classifications11m
- Periodic Table: Group Names8m
- Periodic Table: Representative Elements & Transition Metals7m
- Periodic Table: Elemental Forms (Simplified)6m
- Periodic Table: Phases (Simplified)8m
- Law of Definite Proportions9m
- Atomic Theory9m
- Rutherford Gold Foil Experiment9m
- Wavelength and Frequency (Simplified)5m
- Electromagnetic Spectrum (Simplified)11m
- Bohr Model (Simplified)9m
- Emission Spectrum (Simplified)3m
- Electronic Structure4m
- Electronic Structure: Shells5m
- Electronic Structure: Subshells4m
- Electronic Structure: Orbitals11m
- Electronic Structure: Electron Spin3m
- Electronic Structure: Number of Electrons4m
- The Electron Configuration (Simplified)22m
- Electron Arrangements5m
- The Electron Configuration: Condensed4m
- The Electron Configuration: Exceptions (Simplified)12m
- Ions and the Octet Rule9m
- Ions and the Octet Rule (Simplified)8m
- Valence Electrons of Elements (Simplified)5m
- Lewis Dot Symbols (Simplified)7m
- Periodic Trend: Metallic Character4m
- Periodic Trend: Atomic Radius (Simplified)7m
- 3. Ionic Compounds2h 20m
- Periodic Table: Main Group Element Charges14m
- Periodic Table: Transition Metal Charges5m
- Periodic Trend: Ionic Radius (Simplified)5m
- Periodic Trend: Ranking Ionic Radii8m
- Periodic Trend: Ionization Energy (Simplified)9m
- Periodic Trend: Electron Affinity (Simplified)8m
- Ionic Bonding6m
- Naming Monoatomic Cations6m
- Naming Monoatomic Anions5m
- Polyatomic Ions25m
- Naming Ionic Compounds11m
- Writing Formula Units of Ionic Compounds7m
- Naming Ionic Hydrates6m
- Naming Acids18m
- 4. Molecular Compounds2h 18m
- Covalent Bonds6m
- Naming Binary Molecular Compounds6m
- Molecular Models4m
- Bonding Preferences6m
- Lewis Dot Structures: Neutral Compounds (Simplified)8m
- Multiple Bonds4m
- Multiple Bonds (Simplified)6m
- Lewis Dot Structures: Multiple Bonds10m
- Lewis Dot Structures: Ions (Simplified)8m
- Lewis Dot Structures: Exceptions (Simplified)12m
- Resonance Structures (Simplified)5m
- Valence Shell Electron Pair Repulsion Theory (Simplified)4m
- Electron Geometry (Simplified)8m
- Molecular Geometry (Simplified)11m
- Bond Angles (Simplified)11m
- Dipole Moment (Simplified)15m
- Molecular Polarity (Simplified)7m
- 5. Classification & Balancing of Chemical Reactions3h 17m
- Chemical Reaction: Chemical Change5m
- Law of Conservation of Mass5m
- Balancing Chemical Equations (Simplified)13m
- Solubility Rules16m
- Molecular Equations18m
- Types of Chemical Reactions12m
- Complete Ionic Equations18m
- Calculate Oxidation Numbers15m
- Redox Reactions17m
- Spontaneous Redox Reactions8m
- Balancing Redox Reactions: Acidic Solutions17m
- Balancing Redox Reactions: Basic Solutions17m
- Balancing Redox Reactions (Simplified)13m
- Galvanic Cell (Simplified)16m
- 6. Chemical Reactions & Quantities2h 34m
- 7. Energy, Rate and Equilibrium3h 45m
- Nature of Energy6m
- First Law of Thermodynamics7m
- Endothermic & Exothermic Reactions7m
- Bond Energy14m
- Thermochemical Equations12m
- Heat Capacity19m
- Thermal Equilibrium (Simplified)8m
- Hess's Law23m
- Rate of Reaction11m
- Energy Diagrams12m
- Rate Law (Simplified)5m
- Chemical Equilibrium7m
- The Equilibrium Constant14m
- Le Chatelier's Principle20m
- Solubility Product Constant (Ksp)17m
- Spontaneous vs Nonspontaneous Reactions7m
- Entropy (Simplified)9m
- Gibbs Free Energy (Simplified)18m
- 8. Gases, Liquids and Solids3h 27m
- Pressure Units6m
- Kinetic Molecular Theory14m
- The Ideal Gas Law18m
- The Ideal Gas Law Derivations13m
- The Ideal Gas Law Applications6m
- Chemistry Gas Laws17m
- Chemistry Gas Laws: Combined Gas Law12m
- Standard Temperature and Pressure14m
- Dalton's Law: Partial Pressure (Simplified)13m
- Gas Stoichiometry18m
- Intermolecular Forces (Simplified)19m
- Intermolecular Forces and Physical Properties11m
- Atomic, Ionic and Molecular Solids10m
- Heating and Cooling Curves30m
- 9. Solutions4h 27m
- Solutions6m
- Solubility and Intermolecular Forces17m
- Solutions: Mass Percent6m
- Percent Concentrations10m
- Molarity18m
- Osmolarity15m
- Parts per Million (ppm)13m
- Solubility: Temperature Effect8m
- Intro to Henry's Law4m
- Henry's Law Calculations12m
- Dilutions12m
- Solution Stoichiometry14m
- Electrolytes (Simplified)13m
- Equivalents11m
- Molality15m
- The Colligative Properties15m
- Boiling Point Elevation16m
- Freezing Point Depression9m
- Osmosis16m
- Osmotic Pressure10m
- Vapor Pressure Lowering (Raoult's Law)16m
- 10. Acids and Bases3h 10m
- Acid-Base Introduction11m
- Arrhenius Acid and Base6m
- Bronsted Lowry Acid and Base21m
- Acid and Base Strength17m
- Ka and Kb16m
- The pH Scale16m
- Auto-Ionization9m
- pH of Strong Acids and Bases9m
- Acid-Base Equivalents14m
- Acid-Base Reactions7m
- Gas Evolution Equations (Simplified)6m
- Ionic Salts (Simplified)11m
- Buffers11m
- Henderson-Hasselbalch Equation16m
- Strong Acid Strong Base Titrations (Simplified)13m
- 11. Nuclear Chemistry1h 1m
- BONUS: Lab Techniques and Procedures1h 38m
- BONUS: Mathematical Operations and Functions47m
- 12. Introduction to Organic Chemistry1h 34m
- 13. Alkenes, Alkynes, and Aromatic Compounds2h 30m
- Spatial Orientation of Bonds3m
- Intro to Hydrocarbons16m
- Isomers14m
- Chirality15m
- Naming Alkenes11m
- Naming Dienes and Trienes6m
- Naming Alkynes9m
- Intro to Addition Reactions4m
- Halogenation Reaction4m
- Hydrogenation Reaction3m
- Hydrohalogenation Reaction7m
- Hydration Reaction10m
- Naming Benzene19m
- Benzene Reactions10m
- Benzene Reaction: Nitration6m
- Benzene Reaction: Sulfonation5m
- 14. Compounds with Oxygen or Sulfur1h 22m
- 15. Aldehydes and Ketones1h 1m
- 16. Carboxylic Acids and Their Derivatives1h 11m
- 17. Amines40m
- 18. Amino Acids and Proteins2h 2m
- 19. Enzymes1h 37m
- 20. Carbohydrates1h 46m
- Intro to Carbohydrates4m
- Classification of Carbohydrates4m
- Fischer Projections4m
- Enantiomers vs Diastereomers7m
- D vs L Enantiomers9m
- Cyclic Hemiacetals8m
- Intro to Haworth Projections4m
- Cyclic Structures of Monosaccharides11m
- Mutarotation4m
- Reduction of Monosaccharides10m
- Oxidation of Monosaccharides7m
- Glycosidic Linkage14m
- Disaccharides7m
- Polysaccharides8m
- 21. The Generation of Biochemical Energy2h 9m
- 22. Carbohydrate Metabolism2h 31m
- 23. Lipids2h 26m
- Intro to Lipids6m
- Fatty Acids25m
- Physical Properties of Fatty Acids6m
- Waxes4m
- Triacylglycerols12m
- Triacylglycerol Reactions: Hydrogenation8m
- Triacylglycerol Reactions: Hydrolysis13m
- Triacylglycerol Reactions: Oxidation7m
- Glycerophospholipids15m
- Sphingomyelins13m
- Steroids15m
- Cell Membranes7m
- Membrane Transport10m
- 24. Lipid Metabolism1h 45m
- 25. Protein and Amino Acid Metabolism1h 37m
- 26. Nucleic Acids and Protein Synthesis2h 54m
- Intro to Nucleic Acids4m
- Nitrogenous Bases16m
- Nucleoside and Nucleotide Formation9m
- Naming Nucleosides and Nucleotides13m
- Phosphodiester Bond Formation7m
- Primary Structure of Nucleic Acids11m
- Base Pairing10m
- DNA Double Helix6m
- Intro to DNA Replication20m
- Steps of DNA Replication11m
- Types of RNA10m
- Overview of Protein Synthesis4m
- Transcription: mRNA Synthesis9m
- Processing of pre-mRNA5m
- The Genetic Code6m
- Introduction to Translation7m
- Translation: Protein Synthesis18m
Functional Group Priorities: Videos & Practice Problems
When naming compounds with multiple functional groups, prioritize the highest-ranking functional group as the parent chain. The order of priority is: carboxylic acids, aldehydes, ketones, alcohols, amines, and ethers. Use specific prefixes for substituents: alkoxy for ethers, amino for amines, hydroxy for alcohols, and oxo for aldehydes or ketones. Remember the mnemonic "cool alligators keep all animals excited" to recall the order of functional groups. This systematic approach is essential for accurately identifying and naming complex organic compounds.
Functional Group Priorities Concept 1
Functional Group Priorities Concept 1 Video Summary

Functional Group Priorities Example 1
Functional Group Priorities Example 1 Video Summary
When determining the parent chain names for organic compounds, it is essential to recognize the functional groups present and their priority. In the first compound, there are two functional groups: an amine and an alcohol. Alcohols have a higher priority than amines, which means the parent chain will be based on the alcohol. To name the alcohol, we start numbering the carbon chain from the end closest to the hydroxyl group (–OH). In this case, the numbering goes as follows: 1, 2, 3, 4, 5, 6, 7. Since the alcohol is on carbon number 1 and there are seven carbons in total, the compound is named heptanol.
In the second compound, we encounter a carboxylic acid and a ketone. Carboxylic acids are the highest priority functional group, so we focus on that for naming the parent chain. The carbon chain is numbered starting from the carboxylic acid end, leading to the following sequence: 1, 2, 3, 4. With four carbons in the chain, the parent name is butanoic acid. Although there are additional substituents, such as a methyl group and a ketone, the primary focus remains on the parent chain derived from the highest priority functional group.
In summary, when naming organic compounds, always identify the highest priority functional group to establish the parent chain, while other groups will be treated as substituents in the final name.
Substituent Prefixes Concept 2
Substituent Prefixes Concept 2 Video Summary
When naming organic compounds, it's essential to recognize how functional groups are designated as substituents. Each functional group has a specific prefix that is used when it acts as a substituent in a larger molecule. For instance, when an ether group is present, it is referred to as an alkoxy group. This means that if the ether portion is represented as OCH3, the methyl group (CH3) becomes methoxy due to the presence of the oxygen atom. Similarly, if the ether portion is an ethyl group, it is termed ethoxy instead of simply ethyl.
Other functional groups also have specific names when they serve as substituents. An amine group is called amino, while an alcohol is referred to as hydroxy. Additionally, if an aldehyde or ketone is present as a substituent, it is designated as oxo. Understanding these naming conventions is crucial for accurately identifying and communicating the structure of organic compounds.
Substituent Prefixes Example 2
Substituent Prefixes Example 2 Video Summary
To determine the systematic name of a molecule, it is essential to prioritize functional groups based on their hierarchy. A useful mnemonic for remembering this order is "Cool Alligators Keep All Animals Excited," which indicates that ketones take precedence over alcohols. In this case, the ketone functional group, represented by a carbonyl (C=O), is given higher priority than the alcohol (hydroxy) group.
When naming the molecule, the numbering of the carbon chain begins from the end closest to the highest priority functional group. For an 8-carbon chain, the numbering would proceed as follows: 1, 2, 3, 4, 5, 6, 7, 8. The hydroxy group, which is a substituent, is located on carbon 7, thus it is referred to as "7-hydroxy." The ketone, with its carbonyl group on carbon 3, is denoted as "3-oxo" (the prefix "oxo" is often used for ketones in IUPAC nomenclature).
Since the molecule consists of an 8-carbon chain, it is classified as an "octane." However, because of the presence of the ketone functional group, the suffix changes from "-ane" to "-one," resulting in "octanone." Therefore, the complete IUPAC name for this structure is "7-hydroxy-3-octanone."
Provide IUPAC name for following.
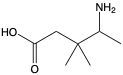
4-amino-3,3-dimethylpentanoic acid
4-amine-3-dimethylpentanoic acid
2-amino-3-dimethylpentaneoic acid
2-amine-3,3-dimethylpentanoic acid
Provide IUPAC name for following.
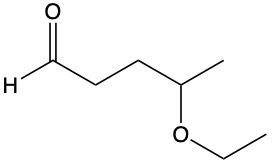
4-propoxypentanal
3-ethoxybutanal
2-ethoxypentanol
4-ethoxypentanal
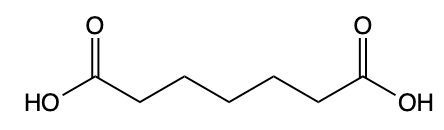
Draw a structure for 7-oxoheptanoic acid.
Do you want more practice?
More setsGo over this topic definitions with flashcards
More setsHere's what students ask on this topic:
When naming organic compounds with multiple functional groups, the order of priority determines which group is named as the parent chain. The highest priority functional group forms the suffix of the compound's name, while others are named as substituents. The priority order is: carboxylic acids > aldehydes > ketones > alcohols > amines > ethers. A helpful mnemonic to remember this sequence is "cool alligators keep all animals excited," where each word corresponds to a functional group in descending priority. This systematic approach ensures consistent and clear naming of complex molecules.
When a functional group is not the highest priority and acts as a substituent, it is named using specific prefixes rather than its usual suffix. For example, ethers become "alkoxy" groups (e.g., methoxy for OCH3), amines are called "amino," alcohols are "hydroxy," and aldehydes or ketones are referred to as "oxo." This naming convention helps clearly indicate the presence of these groups as side chains attached to the main parent structure, maintaining clarity in the compound's systematic name.
Carboxylic acids have the highest priority because they contain multiple functional elements, including a carbonyl group (C=O) and a hydroxyl group (–OH) attached to the same carbon, making them highly reactive and distinctive. This complexity and reactivity justify their precedence in naming. When present, the carboxylic acid group defines the parent chain's suffix ("-oic acid"), ensuring the compound's name reflects the most significant functional group accurately.
The mnemonic "cool alligators keep all animals excited" is a useful tool to remember the priority order of common functional groups in organic chemistry. Each word corresponds to a functional group in descending order of priority: "cool" for carboxylic acids, "alligators" for aldehydes, "keep" for ketones, "all" for alcohols, "animals" for amines, and "excited" for ethers. This phrase helps students quickly recall the correct sequence when naming multifunctional compounds.
When an ether group acts as a substituent, it is named as an "alkoxy" group. This means the alkyl group attached to the oxygen is named first, followed by "-oxy." For example, if the substituent is OCH3, it is called "methoxy." If the substituent is OCH2CH3, it is called "ethoxy." This naming convention distinguishes the ether substituent from the parent chain and clarifies its structure within the molecule.